INTRODUCTION
Since the International Subarachnoid trial, coil embolization of aneurysms is now accepted as a primary optional treatment19). However, endovascular treatment (EVT) of middle cerebral artery (MCA) aneurysms has been less often applied in many institutions because of the feasibility of surgical clipping afforded by the superficial anatomy of MCA aneurysms and favorable outcomes8,24). A large series of EVT of MCA aneurysms was recently reported with favorable results and a low recurrence rate4,11,27), but EVT of wide-necked and fusiform MCA aneurysms remains still challenging. After self-expandable stents became available, many of complex aneurysms have been treated by coil embolization1,3,17,18,21,23). To our knowledge, there are a few reports regarding stent-assisted EVT of MCA aneurysms, but they treated complex MCA aneurysms with one self expandable intracranial stent6,26,29). The aim of this study was to evaluate the feasibility and clinical and angiographic outcomes of stent-assisted coiling for complex MCA aneurysms using more than one stent.
MATERIALS AND METHODS
The institutional review board of our hospital approved this retrospective study, and the requirement for written informed consent was waived. We treated 101 MCA aneurysms with coil embolization in 98 patients between April 2006 and August 2011. Twenty-four aneurysms in 23 patients were treated with stent-assisted coil embolization. Our study involved 13 males and 10 females, and the mean age of the patients was 56.9 years (range, 42-70 years). Nineteen of 24 were incidental aneurysms that had not been treated, and two were recanalized aneurysms previously coiled, and two were recurred after surgical clipping. The number of unruptured aneurysms were 23 and ruptured aneurysm was only one which was ruptured after surgical clipping.
Aneurysm geometry
The mean size of the aneurysms was 7 mm (range, 3-13.4 mm), and mean size of the aneurysm neck was 6.2 mm (range, 2.4-12.7 mm). The mean sac-to-neck ratio (SNR) was 1.02 (range, 0.53-1.45) and the mean height-to-neck ratio was 0.75 (range, 0.48-1.3). All aneurysms had wide neck and/or shallow depths, and six aneurysms were "ultra-wide necked" circumferential aneurysm which is involving more than 180 degree of parent artery lumen with no visible neck. In circumferential aneurysm, size was measured by long dimension and SNR could not be measured. Five aneurysms had incorporated parent vessel branch. There were 17 aneurysms in MCA bifurcation, 3 proximal inferior divisional branch, 2 distal M1, and one proximal superior divisional branch.
Antiplatelet regimen
Patients were premedicated with 75 mg of clopidogrel (Plavix; Bristol-Myers Squibb, New York, NY, USA) and 100 mg of aspirin for 5-7 days. If antiplatelet premedication was not done, on a day before treatment, patients were premedicated with a loading dose of clopidogrel (300 mg) and aspirin (100 mg). After coiling, patients were then kept under dual antiplatelet therapy daily for 6 months (clopidogrel, 75 mg, and aspirin, 100 mg) followed by aspirin daily alone for 12 months. In ruptured aneurysm, antiplatelet premedication was not done and same protocol was applied after procedure.
Procedure of stent-assisted aneurysm treatment
All procedures were performed using a monoplane and biplane angiographic system (Integris V-5000 or Allura Xper, Philips Healthcare, Best, the Netherlands) with the patient under local anesthesia. Anticoagulation was initiated by injection of a bolus of 3000 to 5000 IU heparin intravenously at the beginning of the procedure, followed by continuous infusion of heparin at the rate of 1000 IU/h. Activated coagulation time was maintained between two and three times the baseline value during and for the next 24 to 48 hours after the procedure. In one ruptured aneurysm, a bolus of 3000 IU heparin was injected intravenously after two coils were inserted in the aneurysm, and followed by same heparinization. Endovascular coiling was performed via a single femoral route in all cases with an envoy 6 F (Cordis Endovascular Systems, Miami Lakes, FL, USA) or a shuttle 6 F guiding catheter (Cook, Inc., Bloomington, IN, USA). After completion of coiling, a closing device (Angioseal; St Jude Medical, St Paul, MN, USA, or Perclose; Abott, Diegem, Belgium) was used to seal the access site in all cases.
We initially tried to treat all complex aneurysms using single or double microcatheter or balloon-assisted techniques without stent-assistance. If parent artery or branching vessel is compromised by frame coils or coil frame is unstable, we performed stent-assisted coiling during same procedure.
Fifteen aneurysms were treated using one self-expandable intracranial stent (Neuroform stent, Boston Scientific, Natick, MA, USA; or Enterprise self-expanding stent, Cordis, Miami Lakes, FL, USA). A stent placed into the parent vessel, bridging the aneurysm neck. Stent placement was performed either before coiling (taking advantage of the open cell design of Neuroform stents) or with the jailing technique (using Enterprise stents) when the size of the artery allowed safe navigation of two simultaneous microcatheters. In selected cases, the stent was deployed after coiling at the end of the procedure with the aim of securing the parent artery.
Six aneurysms were treated with a stent-within-a-stent (overlapping two stents) when aneurysm treatment was incomplete with one stent due to very wide neck or fusiform aneurysm for more insertion of coils and effective flow diversion. The stent-within-a-stent technique involves stent-assisted coiling followed by overlapping stent(s). A 0.021-inch microcatheter is navigated across the aneurysm neck portion to a distal branch of the parent artery. A second microcatheter is inserted into the aneurysm sac, and the first Enterprise stent is deployed, bridging the aneurysm neck. After the first stent is deployed, the microcatheter is re-advanced to the initial position over the stent-loading wire, which is left in situ within the deployed stent. When the first deployed stent was Neuroform stent, a micorwire was re-advanced through the deployed stent into the M2 branch and the microcatheter was repositioned in the distal portion of the first deployed stent. Then coil embolization is performed as compactly as possible, including the aneurysm neck portion, which encompasses the parent artery more than 180 degrees. Finally, a second Enterprise stent is introduced and is deployed in an overlapping manner.
Two MCA bifurcation aneurysms were treated with Y-stent if one stent was insufficient for covering the aneurysm neck which was involving both branches. Y-stent-assisted coil embolization was performed as follows. A microcatheter was navigated into the MCA bifurcation aneurysm with or without frame coiling and another microcatheter was placed in the distal superior or inferior divisional branch at the most acute angle from the MCA bifurcation. A 021 Prowler select plus microcatheter was placed at the distal MCA through a long exchangeable wire, and the first stent was deployed from the M2 and the M1 of MCA. The second stent was navigated through the interstices of the initial stent with its distal end in the another bifurcation branch of MCA and the proximal portion overlapping the first stent in the M1 of MCA. Aneurysms were then occluded with coils.
Coiling was performed using Axium coils (EV3, Plymouth, MA, USA), Guglielmi detachable coils and Target coils (Boston Scientific, Natick, MA, USA), HydroCoils (MicroVention, Aliso Viejo, CA, USA), or a combination of coils. In the event of a periprocedural thromboembolic complication, Tirofiban (Aggrastat, Medicure Pharma, Somerset, NJ, USA) was slowly injected intra-arterially as a loading dose (500 mcg/10 mL) and then administered intravenously, at an initial rate of 0.4 mcg/kg/min for 30 minutes and then continued at 0.1 mcg/kg/min.
Patient follow-up
The clinical outcomes of patients were evaluated during hospitalization after procedure and then re-evaluated at the first time of angiographic control, which was generally performed at 6-12 months by DSA. Additional controls by MRA were then scheduled at 2 years, 3 years, and 5 years. In our institution, the MRA sequences (time-of-flight) were analyzed by using source images; maximum-intensity-projection and contrast enhanced MRA images were irrelevant due to major artifacts caused by the stents. In cases of unreliable MR imaging findings, DSA was performed. Morbidity-mortality rates and angiographic results were retrospectively analyzed at initial and midterm follow-ups. We used the Raymond classification scale22) to compare the initial and midterm angiographic results. We defined the midterm results as follows: 1) a stable result showed no change from the initial results; 2) the improved state demonstrated further occlusion of the remnant neck or residual aneurysm; 3) a minor recurrence revealed no need for retreatment (aneurysms changed from the class 1 to class 2); and 4) a major recurrence required requiring retreatment (aneurysms changed from class 1 to class 3 or from class 2 to class 3).
RESULTS
A total of 23 patients having 24 MCA aneurysms were eligible for EVT with the stent assisted coiling technique. Treatment techniques, initial and follow-up angiographic outcomes, and treatment related complication after stent assisted coiling of MCA aneurysms are shown in Table 1.
Treatment techniques
Fifteen aneurysms were treated by the use of a single stent, six aneurysms by a stent-within-a-stent procedure, and two using the Y-stent technique. Twenty-three aneurysms were successfully treated with a stent-assisted technique (95.9%) and one aneurysm embolization was failed due to an inability to catheterize the inferior divisional branch due to acute angle and complex geometry.
Treatment related complications
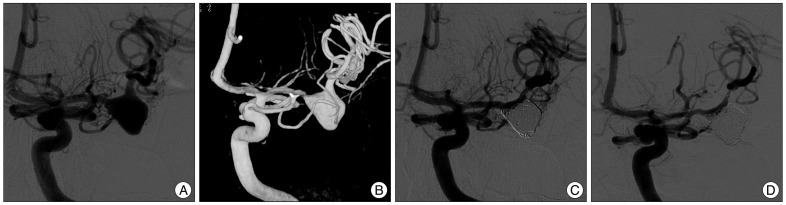
In-stent thrombosis was noted in five patients (20.8%) with symptomatic thromboembolism in two patients, and they showed complete or nearly complete resolution of the thrombi after intra-arterial and intravenous administration of glycoprotein IIb/IIIa inhibitor (Tirofiban, 12.5 to 25 mg). Two symptomatic patients with dysarthria and left side weakness showed complete recovery without permanent deficit. One aneurysm was treated with single stent technique and achieved complete embolization of the MCA bifurcation aneurysm, but the superior divisional branch was occluded due to in-stent thrombosis. Another stent was deployed within the stent after 20 minutes following partial recanalization of the occlusion by Tirofiban injection and immediate control angiogram showed complete recanalization of the superior divisional branch (Fig. 1). Immediate thromboembolic complication was seen in 5 patients (20.8%) during or after procedure showing motor weakness (grade II-IV), dysarthria, and facial weakness. Two symptomatic thromboembolisms were resolved by Tirofiban injection and one left side weakness was disappeared after conservative treatment. Permanent morbidity was noted in 2 patients (8.3%) with partial aphasia and grade III motor weakness [(modified Rankin Scale (mRS) 1 and 2]. Periprocedural thromboembolic complication was noted in 3 single stent-assisted coiling, 1 a stent-within-a-stent technique, and 1 Y-stent technique. Intraprocedural aneurysm rupture occurred in one patient who was treated with a single-stent assisted technique. The rupture was immediately controlled by the insertion of further coils and left no sequelae except for transient post-procedural headaches. No hemorrhagic complication was noted.
Clinical and angiographic follow up
Postembolization control angiograms revealed complete occlusion in 15 (65.2%), residual neck in 5 (21.7%), and a residual aneurysm in 3 (13.1%) patients. Clinical follow-up was available in all patients (mean, 28 months; range, 5-60 months). There were no cases of SAH and no deaths by the recurrence of the coiled aneurysms. None of the surviving 23 patients had any additional deterioration of functional neurological outcomes (mRS, 0-2).
Follow-up angiography was available in 17 of the 23 treated aneurysms (73.9%) by using either MRA (n=5, 29.4%) or DSA (n=12, 70.6%) at a mean period of 13.2 months (6-31 months). An artifact of the stent disturbed MRA interpretation in two cases for which DSA was performed. Fourteen (82.4%) were stable (Fig. 2) and one (5.9%) improved. One major and one minor recurrence were detected. One major recurrent aneurysm treated by the stent-within-a-stent technique was retreated using a stent-assisted technique without any complications (Fig. 3). We thought this aneurysm may have been a dissecting aneurysm.
DISCUSSION
Since the advent of the detachable coil, endovascular coil embolization has been an important treatment method for both ruptured and unruptured aneurysms. However, coil embolization of aneurysms with wide neck and complex geometry remains a challenging procedure despite the use of several techniques such as balloon-assisted20,30), double2) or multiple microcatheter12), microcatheter protective15), coil protective, and stent-assisted techniques1,3,6,7,17,18,21,23,26,29). Ever since the advent of self-expandable intracranial stents for coil embolization of aneurysms, especially closed cell design stent (Enterprise and Leo stent, BALT Co., Montmorency, France), stent-assisted embolization has been widely used due to ease of deployment of the stent and high success rates10). Recently, many of complex aneurysms is treated by stent-assisted technique rather than balloon-assisted and multiple microcatheter techniques. Coil embolization of unruptured MCA aneurysms has favorable outcomes and lower complication rates4,11). Stent-assisted embolization of the complex MCA aneurysms was feasible, safe and durable, and could be considered as an alternative treatment26). However, surgical clipping of MCA aneurysms is preferred in many institutions due to relative ease and effectiveness of aneurysm clipping.
The initial complete or near-complete embolization rate in our series (65.2%) was similar to other studies (67-71%)6,26,29). We performed the jailing technique in 17 aneurysms and repositioning of the microcatheter through the strut of the stent in the remnant neck or aneurysm during procedure if the jailed microcatheter was kickback in the parent artery. A stent-within-a-stent technique in six aneurysms allowed more compact packing and efficient flow diversion than single stent-assisted embolization. Flow diverter might be used, especially in the M1 aneurysm without branching parent vessels, but we could not use them because those stents were not available in our country.
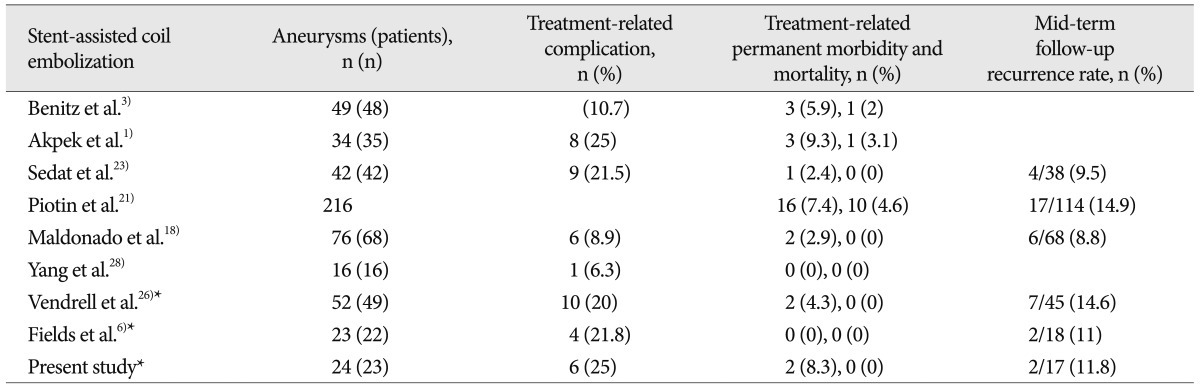
In the present study, in-stent thrombosis (20.8%) and the symptomatic thromboembolic complication rate (20.8%) were high despite the use of dual antiplatelet therapy. We didn't clopidogrel and aspirin resistance test and high thromboembolic complication rate may be antiplatelet resistance. We also think that our initial high complication rate may arise from more complex procedures such as a stent-within-a-stent or Y-stent technique than single stent-assisted technique. The permanent morbidity and mortality rates were decreased as result of complete thrombolysis by immediate Tirofiban injection and leaved two mild disabilities (mRS 1 and 2). However, this morbidity-mortality rate (8.3%) was higher than those of other series (0-12%) (Table 2) and may not be acceptable for treatment method. During recent 3 years, 10 aneurysms was treated and showed no permanent morbidity and one immediate symptomatic complication. In this result, experience of neurointerventionist and careful case selection are important factors for reducing complication rate of stent-assisted coil embolization of the MCA aneurysms.
The recurrence rate for stent-assisted coil embolization of the aneurysms was relatively high in other studies (non-stenting), ranging from 14.6% to 29%4,11,27), and our results were either comparable (11.8%) with previous stent-assisted coil embolization studies (Table 2).
Coil embolization with a stent-within-a-stent technique is a feasible and reconstructive treatment option for ruptured dissecting aneurysms25) and effectively prevents rebleeding and regrowth of ruptured blood blister-like aneurysms without requiring sacrifice of the internal carotid artery14). Therefore, we expected high packing density and lower recurrence rates for large and wide-necked aneurysms while preserving the parent artery, especially ultra-wide necked circumferential MCA aneurysms. In addition to the neck covering role of the stent-assisted technique during embolization, advantages of a stent-within-a-stent technique included prevention of possible protrusion of the coils into the parent artery after first stenting, coil insertion of a very wide-necked circumferential aneurysms due to poor discrimination of the parent artery and neck of the aneurysm due to complex anatomic geometry. Although a down-the-barrel view may help to differentiate the parent artery lumen from coils8), it is not always available as a working projection. Another advantage is prevention of the recurrence of incomplete embolization of the aneurysm due to more effective flow diversion than the single stent, resultant aneurysmal thrombosis, and eventual endothelialization around the neck5,16,28). Finally, blood flow may be compromised due to in-stent thrombosis or coil protrusion within the parent vessel during coiling, so a second stent can easily restore blood flow through the stented parent artery if Tirofiban injection could not recanalized sufficiently (Fig. 1).
Lawson et al.13) reported that stent-assisted coiling causes the progression of occlusion and complete thrombosis of incompletely coiled aneurysms, possibly by a flow remodeling effect, and is therefore associated with lower rates of aneurysm recurrence. There was also a larger series (216 aneurysms) that show a statistically significant lower recurrence rate of stenting (14.9%) compare to coiling (33.5%)21). However, Hwang et al.9) insisted their study did not show that additional hemodynamic and biologic effects of stents designed for neck remodeling were sufficient to enhance progressive occlusion and to prevent the recanalization of unruptured aneurysms. Their findings also suggested that stent placement provides no better long-term angiographic outcomes for unruptured aneurysms with an unfavorable configuration for coiling as compared with nonstent-assisted (multiple microcatheter technique or balloon-assisted technique) coil embolization groups. We believe that selection bias between the stenting and non-stenting groups in each study that were non-randomized and retrospective study, caused the discrepancies in results. We also believe that the most important factor for lower recanalization rate was complete embolization and compact packing of the aneurysms irrespective of the techniques used.
There were several limitations in this study. First, the sample size was too small to conduct a statistical analysis. Another limitation was that we included no comparable non-stenting group or surgical clipping group with same geometry of the aneurysms because unruptured MCAs are not treated by surgical clipping at our hospital. Our limited number of angiographic follow up cases and limited follow up times were another major limitation.
CONCLUSION
MCA aneurysms with wide neck or fusiform could be treated by stent-assisted coiling with more than one stent, and showed good initial angiographic outcomes and lower recanalization rate during mid-term follow-up by effective flow diversion. Our results warrant further study with a longer follow-up period in a larger sample.