Changes in Dermatomal Somatosensory Evoked Potentials according to Stimulation Intensity and Severity of Carpal Tunnel Syndrome
Article information
Abstract
Objective
To investigate the change of latency of cervical dermatomal somatosensory evoked potential (DSEP) according to stimulation intensity (SI) and severity of carpal tunnel syndrome (CTS).
Methods
Stimulation sites were the C6, C7, and C8 dermatomal areas. Two stimulation intensities 1.5×sensory threshold (ST) and 2.5×ST were used on both normal and CTS patients.
Results
In moderate CTS, the latencies of C6 and C7 DSEP during 1.5×ST SI and those of C7 DSEP during 2.5×ST SI were significantly delayed compared with the values of normal subjects. Significant correlation between the latency of C7 DSEP of 2.5×ST stimulation and the median sensory nerve conduction velocity was observed.
Conclusion
We suggest that these data can aid in the diagnosis of cervical sensory radiculopathy using low stimulation intensity and of those who have cervical sensory radiculopathy combined with CTS patients.
INTRODUCTION
The dermatomal somatosensory evoked potential (DSEP) study is a method for evaluating the abnormality of the somatosensory tract, which extends from the peripheral nerve to the cerebral cortex. In 1947, Dawson4) first reported the evoked potentials in humans. Since then, it has been reported to be effective in identifying lesions and then determining their locations in various damage sites, such as peripheral neuropathies, radiculopathies, spinal cord injuries, central nervous system disorders, and cerebral damage1,5,12).
In patients with radiculopathy, however, DSEP remains an auxiliary diagnostic regimen secondary to needle electromyography (EMG). Conflicting opinions about DSEP's diagnostic value in sensory cervical radiculopathy10,19) indicate that more study is needed to optimize its usefulness as a diagnostic procedure. Thus, we conducted a preliminary study in patients with lumbosacral radiculopathy, according to which the sensitivity of DSEP could be enhanced when a stimulus was exerted at a lower intensity compared with conventional intensity13).
Given the above background, we have assumed that low-intensity DSEP might be clinically useful in making a diagnosis of cervical radiculopathy. In this report, we measured the latencies and amplitudes of DSEP depending on the dermatomes of cervical nerve roots and stimulation intensities in normal healthy people. In addition, we performed the DSEP study for patients with carpal tunnel syndrome (CTS), one of the most common neuropathies in the upper extremities. The aim of this study is to acquire reference data which can be helpful for the diagnosis of cervical sensory radiculopathy in patients with or without CTS.
MATERIALS AND METHODS
Study subjects
In this study, a low-intensity DSEP study was performed for 28 hands of 28 normal healthy people and 29 hands of 19 patients who were diagnosed with CTS. The CTS patients were confined to ones who visited Department of Rehabilitation Medicine in this hospital between April 2009 and May 2011. All participants gave informed consent. There were 3 hands which belonged to severe CTS subgroup were discarded from the study. Because they were small in number for statistical analysis, 29 hands of 18 CTS patients were used.
Methods
An electrodiagnostic test was performed using Keypoint® (Medtronics, Denmark), for which the motor nerve conduction study (MNCS) and the sensory nerve conduction study (SNCS) of the median nerve and the ulnar nerve were carried out. Antidromic SNCS was performed for the median nerve and the ulnar nerve; during the test, the hand temperature was maintained at >33℃.
For the MNCS of the median nerve, an active recording electrode was placed on the abductor pollicis brevis, a reference recording electrode was placed on its tendon close to the metacarpophalangeal joint, and a stimulating electrode was placed on the wrist joint, 8 cm proximal to the active recording electrode. For the MNCS of the ulnar nerve, an active recording electrode was placed on the abductor digiti minimi, a reference recording electrode was placed on its tendon close to the metacarpophalangeal joint of the fifth finger, and a stimulating electrode was placed on the wrist joint, 8 cm proximal to the active electrode.
For the SNCS of the median nerve, an active recording electrode was placed on the proximal phalanx of the third finger, a reference recording electrode was placed on the distal phalanx of the third finger, and a stimulating electrode was placed on the palmar region, 7 cm proximal to the active electrode, and 14 cm proximal to the active recording electrode. For the SNCS of the ulnar nerve, an active recording electrode was placed on the proximal phalanx of the fifth finger, a reference recording electrode was placed on the distal phalanx of the fifth finger, and a stimulus was exerted on the wrist, 14 cm proximal to the active recording electrode. The latency, amplitude, and conduction velocity were measured at each session of the test, which was followed by electrodiagnostic severity classification based on the criteria by Steven14).
The DSEP testing was performed using a Keypoint® (Medtronics, Denmark), for which the intensity of stimuli was set at 1.5 times and 2.5 times the sensory threshold. The sweep velocity, sensitivity, filtering selection, stimulation prevalence, stimulation persistence time, and stimulation frequency were set at 10 ms/division, 1 uV/division, 10-3000 Hz, 3 Hz, 0.1 msec, and 200 times, in the corresponding order. Encephalographic needle electrodes were used to record DSEP. Active recording electrodes were placed on C3' or C4' based on the 10-20 international encephalographic system according to the examined hands. A reference electrode was placed on Fz, and a ground electrode was placed between the stimulus electrode and the recording electrode. A stimulating electrode was placed on the proximal phalanx of the thumb for the sixth cervical dermatome, on the proximal phalanx of the third finger for the seventh cervical dermatome, and on the proximal phalanx of the fifth finger for the eighth cervical dermatome with the use of a surface electrode3). Patients were recommended to assume a relaxed posture if possible. The nerve conduction and dermatomal SEP studies were assessed by different testers.
Statistical analysis
Statistical analysis was performed using SPSS 12.0 for Windows. An inter-group difference between normal healthy people and patients was analyzed using a non-parametric method, the Mann-Whitney U test. The difference between the cervical dermatomes was analyzed with a non-parametric method, the Kruskal-Wallis test. Additionally, a detailed comparison was also made for the difference depending on the electrodiagnostic severity of CTS. Furthermore, an analysis was performed to identify the correlation between the sensory nerve conduction velocity and the latency of DSEP with the use of Spearman's test. A value of p<0.05 was considered statistically significant.
RESULTS
General characteristics of the subjects
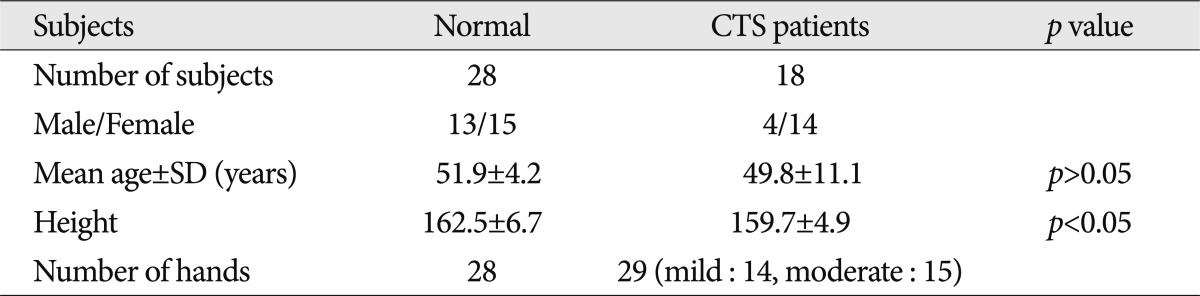
The normal control group comprised 28 normal healthy people, 13 men and 15 women, and the mean age was 51.9 years (range 43-63 years). Patients group of CTS was composed of 4 men and 14 women, and the mean age was 48.9 years (range 29-77 years). There was no significant difference in the age between the two groups. The mean height of normal control group was 162.5 and those of patients group of CTS was 159.7. There was no significant difference in the age between the two groups (Table 1). Of 18 patients with CTS, seven patients had a unilateral presence of lesions, and 11 patients had bilateral lesions. Therefore, a total of 29 lesions were examined in the current study (Table 1).
DSEP study in normal healthy people
In cases of stimulus intensities of 1.5 times and 2.5 times the sensory threshold (ST), a discrepancy was observed in the response rate of DSEP. In cases of stimulus intensity of 1.5 times ST, the response rate was found to be 43% in the sixth cervical dermatome, 61% in the seventh cervical dermatome, and 68% in the eighth cervical dermatome. In cases of stimulus intensity of 2.5 times ST, it was found to be 100%, 100%, and 96%, in the corresponding order.
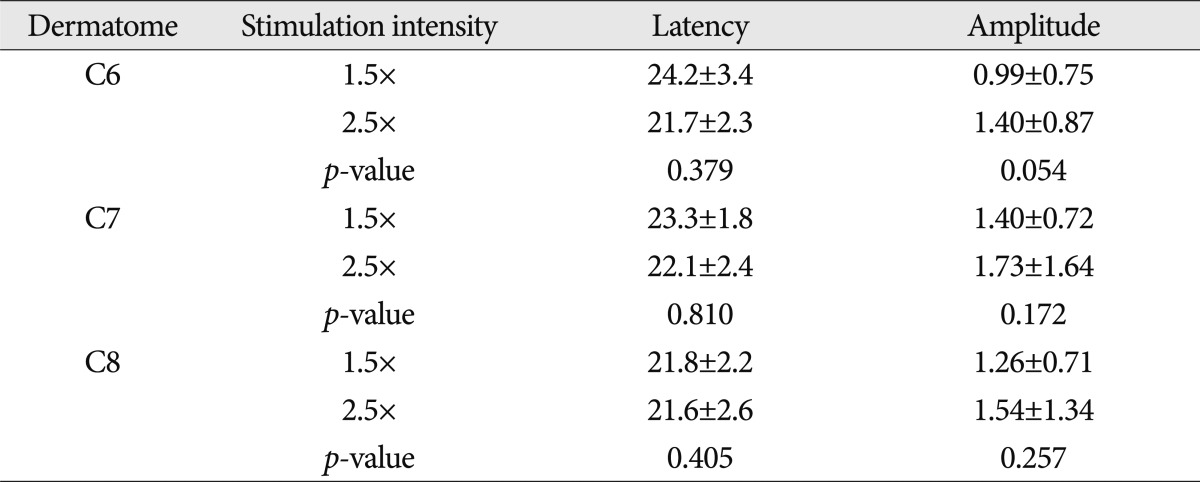
In cases of stimulus intensity of 1.5 times ST, the latency of DSEP was 23.3±1.6 ms in the sixth cervical dermatome, 23.5±1.3 ms in the seventh cervical dermatome, and 23.8±2.0 ms in the eighth cervical dermatome. In cases of stimulus intensity of 2.5 times ST, the latency was found to be 23.0±1.7 ms, 23.1±1.4 ms, and 22.5±1.5 ms, in the corresponding order. No significant difference in latency was found between the cervical dermatomes. However, in the eighth cervical dermatome, the latency was significantly shorter in cases of 2.5 times ST as compared with those of 1.5 times ST (p<0.05).
In cases of stimulus intensity of 1.5 times, the amplitude was 0.95±0.44 µV in the sixth cervical dermatome, 0.97±0.70 µV in the seventh cervical dermatome, and 0.98±0.75 µV in the eighth cervical dermatome. In cases of stimulus intensity of 2.5 times, it was 1.31±0.90 µV, 1.43±0.93 µV and 1.39±0.82 µV, in the corresponding order. These results indicate that no significant difference was seen in the amplitude between the cervical dermatomes. In the seventh and eighth cervical dermatomes, the amplitudes of DSEP were significantly increased in cases of 2.5 times ST compared with those of 1.5 times ST (p<0.05) (Table 2).
DESP study in patients with carpal tunnel syndrome
Using an electrodiagnostic test, 14 mild and 15 moderate cases of CTS were found in the 29 affected hands tested in this study. The mean age of the patients was 50.0±12.0 years and 51.8±9.8 years for mild and moderate cases, respectively. No significant difference in the mean age between the mild and the moderate group was found.
In the affected hands of patients with CTS, there was also a significant difference in the response rate of DSEP according to the stimulation intensity. In cases of stimulus intensity of 1.5 times, the response rate was found to be 55% in the sixth cervical dermatome, 55% in the seventh cervical dermatome, and 76% in the eighth cervical dermatome. In cases of stimulus intensity of 2.5 times, the response rate was found to be 93%, 93%, and 100%, in the corresponding order.
In mild CTS patients, there were no significant differences in the latency depending on the cervical dermatome level and stimulation intensity. In addition, no significant differences were seen in the amplitude depending on the cervical dermatome level and stimulation intensity (Table 3).
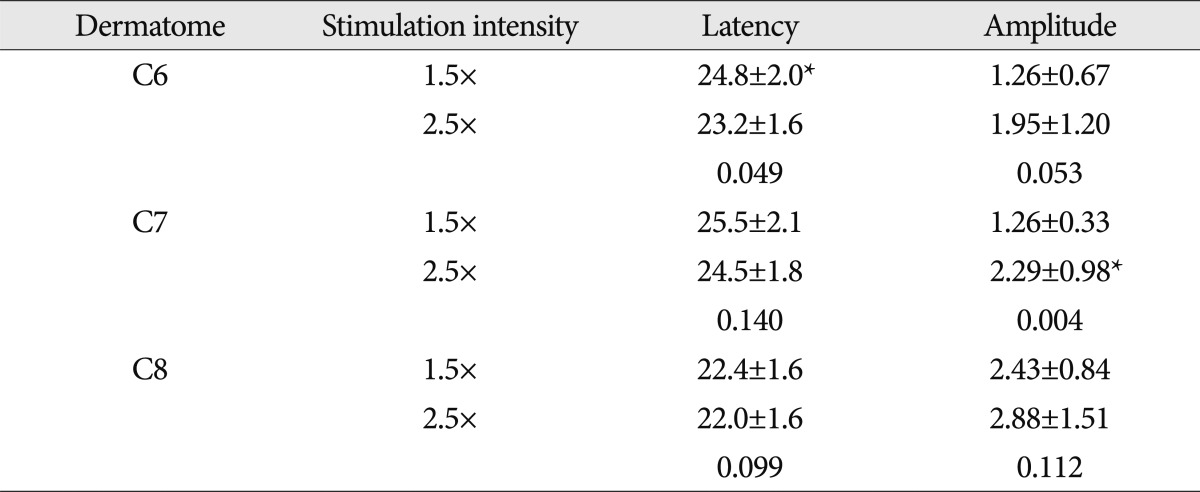
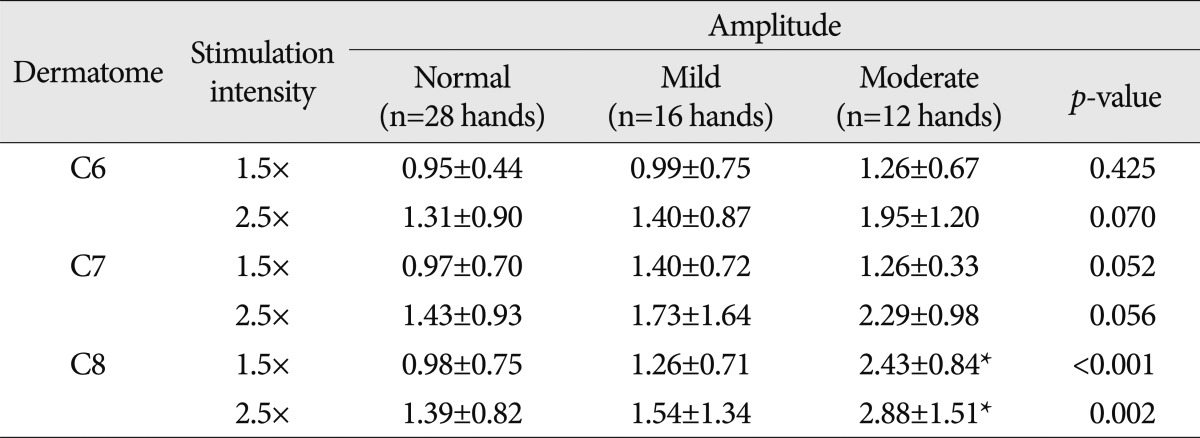
In moderate CTS patients, the latency of DSEP evoked by stimulus intensity of 1.5 times ST was found to be 24.8±2.0 ms in the sixth cervical dermatome, 25.5±2.1 ms in the seventh cervical dermatome, and 22.4±1.6 ms in the eighth cervical dermatome. With the stimulus intensity of 2.5 times ST, the latency was found to be 23.2±1.6 ms, 24.5±1.8 ms, and 22.0±1.6 ms, in the corresponding order. In the sixth cervical dermatome, the latency was more prolonged with the stimulation intensity of 1.5 times ST than with that of 2.5 times ST (p<0.05). Using the stimulus intensity of 1.5 times ST, the amplitude was 1.26±0.67 µV in the sixth cervical dermatome, 1.26±0.33 µV in the seventh cervical dermatome, and 2.43±0.84 µV in the eighth cervical dermatome. Using the stimulus intensity of 2.5 times ST, the amplitude was found to be 1.95±1.20 µV, 2.29±0.98 µV, and 2.88±1.51 µV, in the corresponding order. These results indicate that the amplitude was significantly higher with a stimulus intensity of 2.5 times ST as compared with that of 1.5 times ST in the seventh cervical dermatome (p<0.05). In case of the eighth cervical dermatome, the latency was more prolonged and the amplitude was lower tendency with the stimulation intensity of 1.5 times ST than with that of 2.5 times ST (Table 4).
Comparison of the results of DSEP between normal healthy people and patients with carpal tunnel syndrome
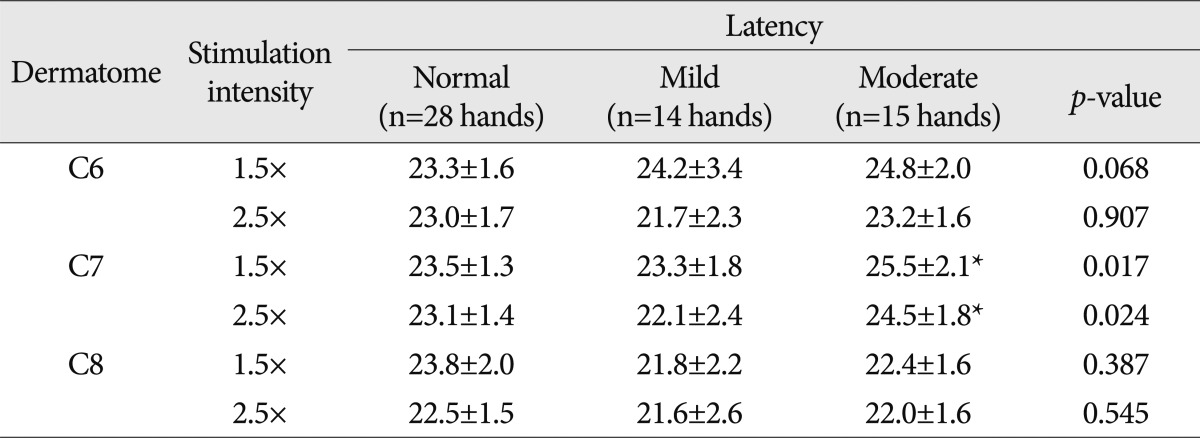
Between normal healthy people and patients with mild or moderate CTS, the latencies and the amplitudes of DSEP were compared. In the seventh cervical dermatome, with the stimulus intensity of 1.5 times and 2.5 times ST, the latency was significantly prolonged in patients with moderate CTS as compared with normal healthy people and patients with mild CTS (p<0.05) (Table 5).
In the eighth cervical dermatome, with the stimulus intensity of 1.5 times and 2.5 times ST, the amplitude of DSEP was significantly higher in patients with moderate CTS as compared with normal healthy people and patients with mild CTS (p<0.05) (Table 6).
The correlation between the sensory nerve conduction study and the DSEP in the patients with carpal tunnel syndrome
In the seventh cervical dermatome with the stimulus intensity of 2.5 times ST, the latency of DSEP had a significant correlation with the conduction velocity of the median sensory nerve in patients with CTS (r2=0.2570, p<0.05). This correlation was evident in patients with moderate CTS (r2=0.4251, p<0.05) (Fig. 1).
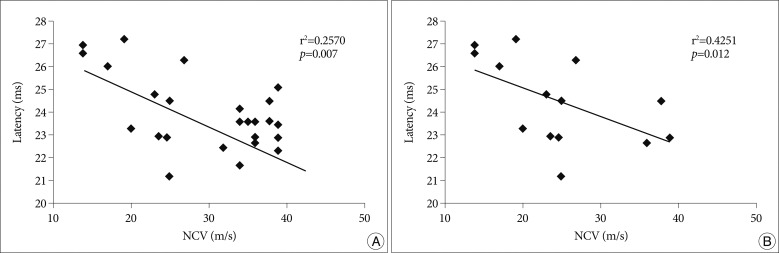
Significant correlation is noted between latency of C7 DSEP and sensory nerve conduction velocity (NCV) in CTS patients, (A) in total CTS patients, (B) in moderate CTS patients, during 2.5×sensory threshold intensity. r2 : Correlation coefficient, p : p-value. DSEP : dermatomal somatosensory evoked potential, CTS : carpal tunnel syndrome.
DISCUSSION
Currently, the reference data for the dermatomal somatosensory evoked potentials in CTS do not exist. Cervical radiculopathy is sometimes combined with the CTS8) When they are combined, it is difficult to investigate how CTS could influence on cervical radiculopathy in dermatomal somatosensory evoked potentials study. To enhance the diagnosis of cervical radiculopathy combined with CTS, we need the reference data of dermatomal somatosensory evoked potentials in CTS patients according to severity. Then, we can define the effect of CTS on cervical radiculopathy during dermatomal somatosensory evoked potentials study. Thus, one of the purpose for this study is to present reference data of CTS accompanied by cervical radiculopathy. We performed the dermatomal somatosensory evoked potentials study not at patients with cervical radiculopathy but at normal healthy people. The control group consisted of patients with CTS.
Tsai et al.18) tried to detect nerve root injuries using a DSEP study in rats, and he reported that the sensitivity and specificity of the DSEP study was higher with submaximal stimulation intensity than with supramaximal stimulation intensity.. In our previous study13), we also reported that the diagnostic sensitivity of DSEP in lumbar radiculopathy patients could be enhanced by lowering the intensity of electrical stimulation as compared with the conventional intensity. Similar significance can be obtained in cervical radiculopathy patients. Before conducting to define the clinical significance of a cervical DSEP study with low stimulation intensity, we suggested to present the normal reference values of cervical DSEP and the changes according to CTS severity, which is one of the most common entrapment syndromes.
In the current study, we performed the cervical DSEP study at a conventional intensity (2.5 times ST) and low intensity (1.5 times ST) for the sixth, seventh, and eighth cervical dermatomes in normal healthy people and patients with CTS. This study showed that the response rate was relatively lower with the low stimulation intensity than with the conventional intensity. In performing the DSEP study, the conventional intensity of stimulation is 2.5 times ST. This is based on the report made by Katifi and Sedwick7), in which the amplitude of DSEP reached approximately 80% of the maximum amplitude with a stimulation intensity of 2.5 times ST. These authors also noted that there was no further decrease of the latency of DSEP at a higher intensity of 2.0 times ST. In the current study, the latencies of DSEP using 2.5 times ST was relatively decreased in comparison with those of 1.5 times ST in normal controls, with statistical significance only in the eighth cervical dermatome. Moreover, the amplitudes of DSEP were significantly increased with 2.5 times ST with statistical significance in the seventh and the eighth dermatomes.
The somatosensory evoked potential study is a method that mainly evaluates the function of large myelinated sensory fibers of the posterior column in the spinal cord. The DSEP study has been designed to evaluate sensory nerve fibers of related to a specific root2). The better diagnostic sensitivity achieved by use of low stimulation intensity may be due to the activation of a fraction of the stimulated dermatomal sensory fibers, so that the function of problematic sensory fibers will be exposed better in a DSEP study when compared to the cases in which all dermatomal sensory fibers are activated. In other words, a DESP study using strong conventional stimulation intensities will activate more dermatomal sensory fibers and the focal problem of sensory fibers will be overlooked.
The DSEP test has been used to help determine the location of lesions in patients with spinal cord diseases or sensory radiculopathy, and it has also been used to make a differential diagnosis between sensory radiculopathy and peripheral neuropathy in patients who complained of severe subjective pain but had no abnormal findings on the conventional EMG11). The DSEP test could be helpful for determining the severity and the prognosis of diseases according to a study which was conducted in patients with peripheral nerve injuries6).
In the current study, the latencies of DSEP in the moderate CTS were increased significantly, especially at stimulation of the sixth and seventh cervical dermatomes compared to the normal controls. This is because the nerve fibers related to the DSEP of the sixth and seventh cervical dermatomes passes through the carpal tunnel and are affected by the entrapment. However, the latencies of DSEP in the mild CTS were not changed significantly compared to the normal controls. These results will be helpful in defining the abnormality of DSEP in the patients who are suspected of sensory cervical radiculopathy and CTS.
In the DSEP study of eighth cervical dermatomes of the CTS patients, the amplitudes were significantly increased as compared with the other cervical dermatomes and with the normal control or the mild CTS patients. Tinazzi et al.17) reported neuroplasticity at multiple levels of the somatosensory system in patients with CTS. The median nerve injury leads to altered afferent processing throughout the somatosensory system, and the ulnar nerve SEP changes. It is thought to be caused by the changes in synaptic strength and unmasking inputs secondary to disconnection of the normally dominant inputs to the median nerve cortex. Also, Napadow et al.9) reported somatosensory cortical plasticity in CTS using functional magnetic resonance imaging evaluation. The ulnar nerve innervated cortex showed a trend for greater activation in CTS patients as compared to healthy adults. Referring to the previous studies, neural plasticity induced activation of the eighth cervical dermatomal pathway to the cortex and the amplitude of the eighth cervical dermatome was increased.
Additionally, using the stimulus intensity of 1.5 times and 2.5 times ST, the latency was significantly prolonged in patients with moderate CTS as compared with normal healthy people and patients with mild CTS in the seventh dermatome but not the sixth dermatome. We believe that it is because sensory fibers of the median nerve have a funicular distribution inside the carpal tunnel, where funiculi coming from the digital nerve of the third finger are more medially located than in the second finger15,16) So, they are presumably more susceptible to damage from intrafunicular anoxia in the condition of CTS.
According to the findings that the amplitudes are affected significantly in the moderate CTS patients but not in the mild CTS patients, it is presumed that the difference in the neural plasticity might be subject to the severity of CTS. In this study, the severe CTS group was discarded because of robust statistical analysis was not possible on the small number of patients available. The relatively low response rate especially in the DSEP study stimulated with 1.5 times ST can be a limitation in clinics. To draw reliable, generalized conclusions about the DSEP study with low stimulation intensity used in this study, further large-scale studies will be necessary.
CONCLUSION
In the current study, we examined the normal controls for the reference data of DSEP depending on the stimulation intensity. In addition, we also reported the changes of DSEP according to the severity of CTS and the stimulus intensities. Through this study, we expect to present the reference data when we perform the DSEP study after cervical radiculopathy is diagnosed. This reference also is expected to be the preliminary data for studies on low intensity stimulation DSEP. Furthermore, these data can be helpful in the diagnosis of sensory cervical radiculopathy combined with CTS.
Acknowledgements
This paper was supported by research funds of Chonbuk National University in 2009.