Deep Intracerebral Hemorrhage Caused by Rupture of Distal Lenticulostriate Artery Aneurysm : A Report of Two Cases and a Literature Review
Article information
Abstract
Intracerebral hemorrhage (ICH) is common among various types of storkes; however, it is rare in young patients and patients who do not have any risk factors. In such cases, ICH is generally caused by vascular malformations, tumors, vasculitis, or drug abuse. Basal ganglia ICH is rarely related with distal lenticulostriate artery (LSA) aneurysm. Since the 1960s, a total of 29 distal LSA aneurysm cases causing ICH have been reported in the English literature. Despite of the small number of cases, various treatment methods have been attempted : surgical clipping, endovascular treatment, conservative treatment, superficial temporal artery-middle cerebral artery anastomosis, and gamma-knife radiosurgery. Here, we report two additional cases and review the literature. Thereupon, we discerned that young patients with deep ICH are in need of conventional cerebral angiography. Moreover, initial conservative treatment with follow-up cerebral angiography might be a good treatment option except for cases with a large amount of hematoma that necessitates emergency evacuation. If the LSA aneurysm still persists or enlarges on follow-up angiography, it should be treated surgically or endovascularly.
INTRODUCTION
Hemorrhagic stroke constitutes around 10% to 15% of all strokes14). Generally, lobar intracerebral hemorrhage (ICH) is known to be caused by cerebral amyloid angiopathy, while deep ICH is often associated with hypertension41). However, research has indicated that abnormal vascular lesions may lead to deep ICH : arteriovenous malformation, moyamoya disease, aneurysm, and vasculitis. Among vascular lesions, rupture of the distal lenticulostriate artery (LSA) aneurysm is rare. Accordingly, considering the rarity of deep ICH caused by rupture of the distal LSA aneurysm, we report two additional cases thereof and review treatment strategies previously mentioned in the literature pertaining to the specific disease.
CASE REPORT
Case 1
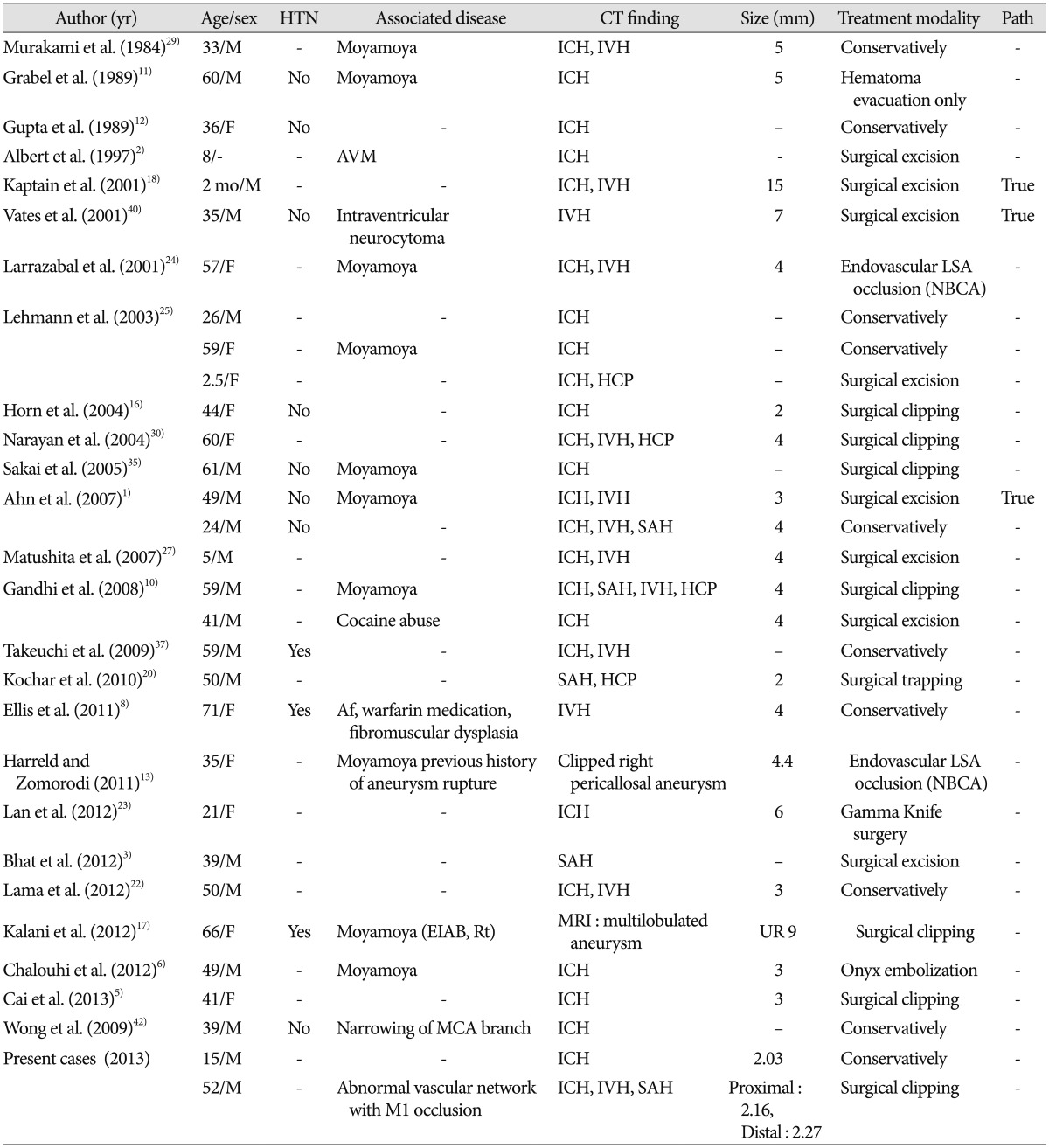
A 15-year-old man presented with a history of right side hemiparesis and dysphasia. He had been healthy with no connective tissue disease or infectious symptom. His laboratory finding did not reveal any thrombocytopenia, coagulopathy or vasculitis. Initial computed tomography (CT) scan at admission revealed an acute left lateral basal ganglia ICH. CT angiography showed an intact circle of Willis without abnormal vascular structures such as aneurysm, arteriovenous malformations, and moyamoya disease (Fig. 1A). After 2 days, magnetic resonance image (MRI) presented no expansion of hematoma without abnormal signal void and mass lesion adjacent to the hematoma (Fig. 1B). Because the patient was so young with no risk factors, we performed distal subtraction angiography (DSA) to identify its cause. The finding was a left lateral LSA small aneurysm with a size of 1.94×2.03 mm, in the most distal position (Fig. 1C). The patient's neurological symptoms were stable and follow-up brain CT showed no more hemorrhage expansion. Therefore, we decided on a conservative treatment strategy and followed up with angiography.
A : Computed tomography (CT) scan at admission shows left putaminal intracerebral hemorrhage (left), and CT angiography shows any abnormal vascular lesion (right). B : Initial magnetic resonance images (MRI) demonstrate no abnormal signal void in T2WI (left) and enhancing mass lesion adjacent to the hematoma in T1 contrast enhanced image (right). C : Initial left internal carotid artery angiogram shows a small aneurysm (arrows) at the distal lateral lenticulostriate artery (LSA) (left : AP view, right : lateral view). D : Follow up angiography after 2 weeks demonstrates complete disappearance of the distal lateral LSA aneurysm (left : AP view, right : lateral view). AP : anterorposterior.
After 2 weeks, follow-up DSA was performed and demonstrated a complete disappearance of distal LSA aneurysm (Fig. 1D). The patient neurologic symptoms had improved and the patient was discharged.
Case 2
A 52-year-old man presented to our hospital with severe headache, nausea and vomitus. He had a history of asthma for 4 years, yet there was no history of hypertension, antiplatelet or anticoagulant assumption, heart disease, infectious disease, or head injury. At admission, physical examination revealed left side facial palsy (House and Backmann grade 2) and a conscious level of Glasgow Coma Scale (GCS) 14 (eye opening 3, verbal 5, motor 6). Initial CT scan revealed subtle basal cistern subarachnoid hemorrhage, more pronounced on the right, and right basal ganglia (caudate head) intracerebral hematoma with intraventricular extension without acute hydrocephalus (Fig. 2A). CT angiography showed right M1 occlusion with right distal LSA aneurysms, two small aneurysms arising from one parent artery. DSA showed right M1 proximal occlusion with an abnormal arterial network; furthermore, one parent artery of the right lateral LSA formed two small aneurysms (Fig. 2B).

A : Non contrast CT at admission shows subtle basal cistern subarachnoid hemorrhage (left), and right caudate head intracerebral hematoma with intraventricular extension without acute hydrocephalus (right). B : First conventional cerebral angiography (AP view, left) and 3D reconstruction (right). The right M1 proximal trunk is aplastic, and distal flow consists of an abnormal arterial network. Aneurysm occurred in one parent artery of the right LSA, forming two small aneurysms. C : Follow-up cerebral angiography after 2 weeks shows enlargement of the aneurysms. (right : AP view, left : 3D reconstruction). D : Intraoperative cerebral angiography (AP view, left : distal subtraction angiography, right : native image) shows complete obliteration of aneurysmal sac by clips. AP : anteroposterior, LSA : lenticulostriate artery.
Follow-up DSA, after 2 weeks of initial cerebral angiogram, was performed and showed an enlargement of the aneurysms (Fig. 2C). Accordingly, we decided on surgical treatment to avoid rebleeding. In the hybrid operation room, under frameless stereotactic guidance, gentle hematoma evacuation and clipping of the aneurysmal neck was done by a transcortical approach. Intraoperative angiogram confirmed successful aneurysm obliteration (Fig. 2D). Postoperatively, the patient was free of neurological symptoms and demonstrated excellent outcomes (mRS 1).
DISCUSSION
Aneurysms of distal LSA are relatively rare. Since the 1960s, thirty-nine English literatures associated with LSA aneurysm were reported123456789101112131516171819202122232425262729303132333435363738394042). Vargas et al.39) classified these aneurysms into three types based on anatomical location. According to this classification, distal LSA aneurysm is classified as type 3. A total of 56 LSA aneurysms were founded in 39 English literatures. Among them, true distal LSA aneurysms were 29 cases. Table 1 showed a summary of 31 distal LSA aneurysms including our cases12356810111213161718202223242527293035374042). The mean age of patients was 40.2 (2 month to 71 yrs). The male to female ratio was 17 to 13 (unknown 1). A history of hypertension was associated in 3 cases. Most ruptured distal LSA aneurysms occurred as a result of intraparenchymal hemorrhage (25 cases, 80.6%). Thirteen cases (41.9%) were associated with vascular lesions, such as moyamoya disease, probable moyamoya disease, arteriovenous malformation, and abnormal vascular network with M1 occlusion. This review revealed peculiar features of a younger mean age of presentation, no female predominance, and greater rates of underlying vascular lesions compared with other cerebral aneurysms. Therefore, young age with deep intracerebral hemorrhage patients should be suspicious of ruptured distal LSA aneurysm and associated vascular lesions. These patient groups should undergo conventional cerebral angiography. However, 11 patients, equivalent to 35%, were over 50 years old. Among them, 5 patients (16.1%) were over the age of 60. McCormick and Rosenfield28) reported that in 144 autopsy cases of massive brain hemorrhage, 58 patients had acceptable evidence of systemic hypertension, and among them, 21 patients (36%) involved other causes for the hemorrhage independent of the hypertension (aneurysm, angioma, cortical vein/dural sinus thrombosis, leukemia, sepsis with vasculitis, collagen-vascular disease, aplastic anemia with thrombocytopenia, and liver failure). This indicates that deep ICH may be due to distal LSA aneurysmal rupture or other underlying structural abnormalities, although deep ICH is commonly associated with pre-existing hypertension in elderly patients. Therefore, initial vessel study, such as CT angiography or MR angiography, may warrant consideration in patients over 50 years of age and who do not have any risk factors of ICH to detect treatable structural lesions.
Despite the small number of cases, various treatment strategies have been attempted. Surgical clipping or trapping were done in 8 patients, surgical excision in 8, endovascular treatment in 3 (NBCA in 1, Onyx in 2), gamma knife radiosurgery in 1, and only hematoma evacuation in 1. Other 10 patients were treated conservatively. In the surgical group, 3 cases demonstrated true aneurysmal pathology, which was typical saccular aneurysm. In the conservative treatment group, meanwhile, 8 cases showed spontaneous disappearance of the aneurysm. The mechanism for the formation of peripheral aneurysms may be related to hemodynamic stress. Distal peripheral aneurysm that disappeared on the follow-up angiogram may have been a pseudoaneurym resulting from previous rupture of a small artery27).
Distal LSA aneurysm are surgically and endovascularly challenging lesions. Surgically, these aneurysms can be located deep in the striatum, making intraoperative localization difficult. As well, aneurysm sizes can be very small, and fragile in nature. These reasons make it difficult to approach these lesions. Nevertheless, direct surgery is advantageous in that it reduces mass effects by the evacuated hematoma, and preserves parent artery patency by neck clipping in typical saccular aneurysm. Difficulty with intraoperative localization can be overcome by using frameless stereotactic guidance and intraoperative angiogram, as was the case here. Endovascularly, the acute angle and the small diameter of the LSA make selective catheterization of the aneurysm neck difficult. In 3 cases in the endovascular treated group, all three consisted of an occluded parent LSA, but there was no aggravation of neurologic symptoms such as contralateral hemiparesis. Compared with surgical intervention, less neuronal tissue damage is involved with endovascular treatment. Therefore, both surgical and endovascular treatment methods are acceptable treatment options in selected cases of distal LSA aneurysms.
CONCLUSION
Initial conservative treatment with careful follow-up conventional angiography may be warranted in pseudoaneurysmal lesions, whereas direct surgical or endovascular treatment should be considered in typical saccular aneurysms. If the LSA aneurysm still persists or enlarges on follow-up angiography, it should be treated surgically or endovascularly. We carefully suggest that initial conservative treatment with follow-up cerebral angiography might be a good treatment option except for cases with a large amount of hematoma that necessitates emergency evacuation.