Outcomes of Surgical Management of Metopic Synostosis : A Retrospective Study of 18 Cases
Article information
Abstract
Objective
To describe the surgical management and postoperative outcomes in infants with metopic synostosis.
Methods
We conducted a 5 years retrospective chart review of patients who underwent surgical correction of metopic synostosis at two university hospitals in Egypt during the period between June 2014 and June 2019. The study is conducted to 18 children. The type of surgical procedures and postoperative outcomes were assessed in all patients.
Results
Five cases (27.8%) underwent endoscopic-assisted suturectomy, 10 cases (55.6%) underwent craniofacial reconstruction, and three cases (16.6%) underwent open burring of the metopic ridge. Fifteen patients underwent one surgery and three patients (16.6%) who need second operation. Ten patients (55.6%) had class I Whitaker classification.
Conclusion
Regardless of type of surgery, the outcomes of surgical correction of metopic synostosis are excellent with only a few patients require revision or develop major complications.
INTRODUCTION
Craniosynostosis is characterized by the premature and abnormal fusion of suture and can happen as part of a multisystem syndrome or as an isolated defect (nonsyndromic). Eight percent of the patients have familial or syndromic forms [6]. In 1890 Lannelongue made the first surgical procedure for correction of craniosynostosis who supported releasing, but not resecting, the fused suture [22].
After 2 years, Lane illustrated the first strip craniectomy, with the extraction of the fused suture, yet because the child died after the operation, this procedure failed to generate an outcome favored until Mehner in 1921 proved the first successful use of strip craniectomy to remove a fused suture. As a consequence of changes in surgical technique it was difficult to make objective analysis and comparisons [8,32].
Whitaker et al. [30] who illustrated a classification of surgical outcomes which identifies category I as patients in whom no surgery was considered advisable or essential by the surgeon, patient, or family. In category II, soft tissue or minor bone contouring revisions were desired, even they were essentially performed. Category III is defined as patients in whom major secondary osteotomies or bone grafting procedures were required or done. These procedures were not as extensive as the original procedures. Category IV was consist of patients in whom a major craniofacial surgery, duplicating or exceeding the extent of the original surgery, was or would be necessary. After all, that classification system has important restrictions. Categories II, III, and IV all represent patients supposed to need further surgery.
Good or fair results, with residual deformities, but not without additional surgery, are not marked from outstanding results. This classification has been used by McCarthy et al. [23] in his study.
MATERIALS AND METHODS
This study was approved by the Ethical Committee of Department of Neurosurgery, Benha University with IRB approval in November 2020.
Study design, patients & surgical options
We conducted a 5 years retrospective chart review of patients who underwent surgical correction of metopic synostosis at two university hospitals in Egypt during the period between June 2014 and June 2019.
Records of 18 children were retrieved. The diagnosis of metopic synostosis was based on computed tomography (CT) and magnetic resonance imaging (MRI) findings. There were no restrictions regarding the gender or year at the surgery.
Before surgery, all patients underwent full preoperative evaluation including clinical examination, radiologic evaluations, photographic evaluation and fundus examination. Then, the postoperative results were evaluated at 1, 3, 6, and 12 months after surgery and then once a year for up to 5 years. At our institutions, the surgical techniques for metopic synostosis are classified into the following categories.
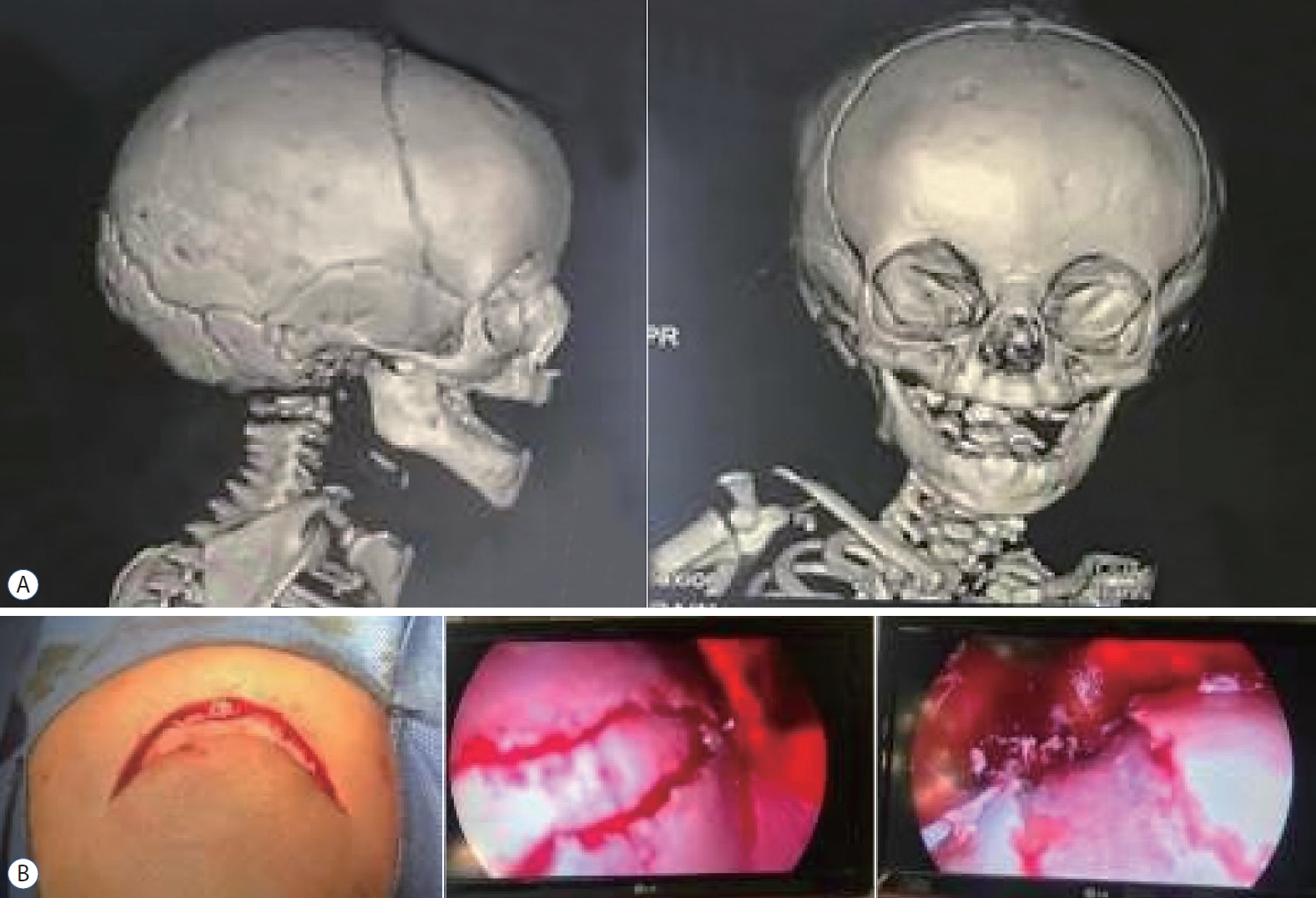
Endoscopic-assisted suturectomy
Minimal invasive craniosynostosis surgeries aim at reducing the morbidity and invasiveness of classical surgical procedures, with equal long-term results, both functional as well as cosmetic, with less intraoperative bleeding and postoperative hospital stay.
Craniofacial reconstruction
This method is used for more complex deformities like when orbital advancement is needed, it involves exposure of the upper eye socket and forehead through an ear-to-ear incision. The forehead bone is then removed, upper eye socket is cut free, reshaped, and replaced in a new position, held in place with resorbable plates and screws. The forehead is then reshaped and affixed to the upper eye socket.
Open burring of the metopic ridge
This method involves metopic suturectomy using the drill and craniotome, metopic suture must be drilled and removed with 2 cm strip craniectomy. The surgical team and the anesthesiology team must work together to reduce risks and treat blood loss quickly to avoid complications.
Case presentation
Case 1
Two years old male child showing skin incision and endoscopic metopic suturectomy with good postoperative results (Fig. 1).
Case 2
Fourteen months old female child operated by craniofacial reconstruction by craniectomy (Fig. 2).
Data collection & study outcomes
We collected the data from every eligible patient : name, age at presentation, gender, age at surgery, nature of deformity, other associated synostosis or abnormalities, surgical procedures, number of surgeries, need for revision surgery, postoperative surgical results, postoperative complications, and inhospital mortality. The postoperative results were assessed according to Whitaker classification six months after surgery (Table 1) [15].
Statistical analysis
The SPSS version 22 for Microsoft Windows (SPSS Inc., Chicago, IL, USA) was used for data processing and analysis. The mean±standard deviation or median with interquartile range were used to describe numerical variables according to the normality of the data. Frequencies with percentages were used to present while categorical variables.
RESULTS
The present study included 18 children who underwent surgical correction of metopic synostosis. The mean age at surgery of the patients was 9.4±2.7 months and 67% of them were males. Besides, the mean age at surgery was 10.1±1.9 months. Five cases had associated synostosis in the form of sagittal (11.1%), coronal (11.1%), or multiple sutures (5.6%). While two patients had associated central nervous system anomalies and one patient had associated cardiac anomalies. The majority of the patients (55.6%) had a deformity of the supra-orbital ridge and temporal region with associated hypotelorism; while the rest of them had only prominent metopic suture.
In terms of surgical procedures, five cases (27.8%) underwent endoscopic technique (ET), 10 cases (55.6%) underwent craniofacial reconstruction, and three cases (16.6%) underwent open burring of the metopic ridge. So the open technique (OT) was 13 patients (72.2%). Estimated blood loss was 55 mL in ET and 150 mL in OT. Operative time was 1.6 hours in ET and 2.8 hours in OT.
Fifteen patients underwent one surgery and three patients (16.6%) who required second surgeries (two were operated by OT and one operated by ET) and follow-up after 6 months show good cosmetic results. The postoperative results are shown in Table 2. Ten patients (55.6%) had class I Whitaker classification. Dural tear occurred in two patients (11.11%) operated with OT. One death (5.6%).
Fundus examination was done routinely for all patients that showed papilledema that resolved in 10 cases (55.55%) in 3 months follow up, resolved in five patients (27.7%) at 6th months, and resolved in three patients (16.7%) at 1 year follow up.
The hospital stay in ET was 2.5 days however the stay was 5 days in OT because we wait for the resolution of postoperative collection. Two patients (11.11%) developed temporal hallowing in OT.
DISCUSSION
This is a retrospective analysis of 18 patients who underwent surgical correction of metopic synostosis at two university hospitals in Egypt between June 2014 and June 2019. Selber et al. [26] recommended early, aggressive, and overcorrection for trigonenecephaly for better aesthetic results.
It is not clear what is the appropriate age to perform the surgery, the mean age at the time of surgery in this study was ten months. Wójcicki and Prudel [32] average age was 18 months. Kelleher et al. [19] median age was 12 months, Keshavarzi et al. [20] median age was 16.6 months, and Engel et al. [11] median age were 11.5 months. Cohen and Persing [8] median age was 8.2 months. Wójcicki and Prudel [32] showed early diagnosis help to perform surgery in the first year of life as surgery at this age provide good result but without correcting behavioral and neurological disorders. Many authors recommended surgical correction during the first 3–9 months [2,9,13].
Hilling et al. [15], patients were operated at a mean age of 13 months (range, 5–51), with 80% having the surgery done between the age of 6 and 15 months. Using Spearman rank correlation, there was no significant correlation between the appearance and age at the time of surgery within the giving range [15]. In the postoperative Whitaker classification, 10 of our patients (55.6%) had a class I outcome, three patients were class II, one was class III, and three patients were class IV. Engel et al. [11] founded that 43 patients (79.6%) had a class I outcome, 10 patients were class II, none of their patients were class III, and only one patient was class IV and frontorbital advancement was the standardized operation. Glener et al. [14] showed fronto-orbital advancement provides immediate and adequate correction in supraorbital bar. Three of our patients (16.7%) need 2nd surgery (two were operated by OT and one operated by ET). However, Nguyen et al. [25] described three patients (8%) who needed a second surgery and were operated by OT and none from ET group. In the assessment of postsurgical relapse, Glener et al. [14] concluded age at intervention and preoperative interfrontal angle (IFA) are the most accurate predictors. However postoperative IFA changes should not be used solely to evaluate relapse due to inherent anatomical limitations. Bhatti-Söfteland et al. [4] found low frontal to total intracranial volume ratio at 3 years of age children was due to under-correction during surgery rather than relapse attributed by ratio was the same at the time of surgery and 3 years old.
Our patients underwent many techniques including endoscopic-assisted suturectomy, craniofacial reconstruction and open burring of the metopic ridge. An endoscopic and minimally invasive technique added potential advantages over the traditional OT [20]. The role of minimally invasive procedures is not clear but the ET is an important treatment option [18]. Early endoscopic approach added important benefits regarding blood loss, blood transfusion and hospital stay [16,17]. Low incidence of venous air embolism reported by Tobias et al. [29].
There is no increased complication in the endoscopic approach but there no significant difference between endoscopic and OTs [20]. Kim et al. [21] reported that the disadvantage of the endoscope that requires banding for 1 year has no bearing on parental perceptions in outcome. Di Rocco et al. [10] described the advantages and disadvantages of the OT and focused on blood loss and large skin incision.
We reported estimated blood loss was 55 mL in ET and 150 mL in OT. Operative time was 1.6 hours in ET and 2.8 hours in OT. Nguyen et al. [25] reported shorter operative time (1.7 hours) in ET and 2.2 hours in OT. 56.8 mL blood loss in ET and 190 mL in OT in Keshavarzi et al. [20] study. Hospital stay in our study in ET group is shorter and similar to other studies and longer in OT as reported by Engel et al. [11] also. This raises the advantages of endoscopic approach with better results and fewer complications. Preoperative fundus examination revealed papilledema in all our patients that resolve in follow up in line with that reported in Engel et al. [11]. Other studies reported signs of high intracranial tension in CT and MRI not presented in our study [8]. Other study provide intracranial pressure (ICP) measurements to support clinical findings in selected cases [27,28] but we did fundus examination as an invasive technique. Volume measurements by CT alone didn’t provide a reliable predictor for elevated ICP [12]. Improvement of behavioral changes noted with the resolution of papilledema [5,7].
We reported dural tear in two cases (11.11%) in the patients operated by OT similar to Engel et al. [11] who reported six patients developed dural tear (11.11%). Other studies reported that the dural tear is more in open techniques [1], and a higher percentage (22%) that presented by other study [24]. The literatures reported rare development of leptomeningeal cyst [3,31]. Two patients (11.11%) developed temporal hallowing in OT compared to other studies that stated (18%) of temporal hallowing [24].
Our study is limited by small sample size, with only 18 cases included in the analysis. We recommend larger studies to address these issues with a larger sample size.
CONCLUSION
Metopic synostosis varies from mild to severe. Mild forms can be treated with metopic ridge burring alone. Severe forms require craniofacial reconstruction and may be associated with developmental delay, additional synostosis, and other congenital abnormalities.
In all cases, the operative decision must be made according to the nature and severity of the deformity. The relation between the bony deformity and its correction and cerebral development requires further study.
Notes
No potential conflict of interest relevant to this article was reported.
INFORMED CONSENT
Informed consent was obtained from all individual participants included in this study.
AUTHOR CONTRIBUTIONS
Conceptualization : MG
Data curation : MEE
Formal analysis : MA
Funding acquisition : MEE, MG
Methodology : MA, MG
Project administration : MEE, MG
Visualization : MG
Writing - original draft : MG, MEE
Writing - review & editing : MG