Moon, Jang, Han, Jang, Huh, and Park: Association Factors for CT Angiography Spot Sign and Hematoma Growth in Korean Patients with Acute Spontaneous Intracerebral Hemorrhage : A Single-Center Cohort Study
Abstract
Objective
This study was conducted to clarify the association factors and clinical significance of the CT angiography (CTA) spot sign and hematoma growth in Korean patients with acute intracerebral hemorrhage (ICH).
Methods
We retrospectively collected the data of 287 consecutive patients presenting with acute ICH who arrived within 12 hours of ictus. Baseline clinical and radiological characteristics as well as the mortality rate within one month were assessed. A binary logistic regression was conducted to obtain association factors for the CTA spot sign and hematoma growth.
Results
We identified a CTA spot sign in 40 patients (13.9%) and hematoma growth in 78 patients (27.2%). An elapsed time to CT scan of less than 3 hours (OR, 5.14; 95% CI, 1.76-15.02; p=0.003) was associated with the spot sign. A CTA spot sign (OR, 5.70; 95% CI, 2.70-12.01; p<0.001), elevated alanine transaminase (GPT) level >40 IU (OR, 2.01; 95% CI, 1.01-4.01; p=0.047), and an international normalized ratio Ōēź1.8 or warfarin medication (OR, 5.64; 95% CI, 1.29-24.57; p=0.021) were independent predictors for hematoma growth. Antiplatelet agent medication (OR, 4.92; 95% CI, 1.31-18.50; p=0.019) was significantly associated with hematoma growth within 6 hours of ictus.
Conclusion
As previous other populations, CTA spot sign was a strong predictor for hematoma growth especially in hyper-acute stage of ICH in Korea. Antithrombotics medication might also be associated with hyper-acute hematoma growth. In our population, elevated GPT was newly identified as a predictor for hematoma growth and its effect for hematoma growth is necessary to be confirmed through a further research.
Key Words: Intracerebral hemorrhage ┬Ę CT angiography ┬Ę Spot sign ┬Ę Hematoma growth ┬Ę Antithrombotics ┬Ę Alanine transaminase.
INTRODUCTION
Spontaneous intracerebral hemorrhage (ICH) is the second most common cause of stroke, occurring globally at a rate of 24.6 per 100000 person-years and causing severe disability and high mortality. In spite of recent clinical trials aimed at improving the outcome of ICH patients, the median case fatality has not decreased over time according to a recent meta-analysis 1,15,27,28,38). CT angiography (CTA) spot sign refers to one or more foci with contrast enhancement within an ICH as a radiologic surrogate marker for hematoma expansion and clinical outcome in acute ICH 11). Hematoma growth is also one of the major determinants of mortality and poor outcome in acute ICH 6,8). Several factors associated with the CTA spot sign have been reported in the Western population, such as large hematoma volume, anticoagulation, and the APOE ╬Ą2 allele 3,4,12). Oral anticoagulant medication, hemostatic proteins, a CTA spot sign, and high systolic blood pressure have also been reported as contributing factors to hematoma growth in acute ICH 1,11,14,26,34). While the incidence of ICH varies among different countries and is relatively high in the Asian population 16,31), there are few studies investigating risk factors and the relationship of the CTA spot sign to hematoma growth in Asian countries 25,32,40). The current study was conducted to investigate the predictive factors associated with the CTA spot sign and hematoma growth of acute spontaneous ICH patients in Korea.
MATERIALS AND METHODS
Populations and study design
From 2005-2012, we collected retrospective data for consecutive patients with spontaneous supratentorial ICH that were admitted to the Stroke and Cerebrovascular Center at our hospital, which serves a population of approximately 560000. The institutional review boards at our hospital approved the current study. The inclusion criteria were the following : 1) age Ōēź18 years old and 2) CTA-proven spontaneous supratentorial hemorrhage. Patients with a cerebral aneurysm rupture, traumatic ICH, tumor bleeding, arteriovenous malformation-related ICH, dural arteriovenous fistula associated ICH, cavernous hemangioma, Moyamoya disease, Moyamoya syndrome, probable Moyamoya disease, venous thrombosis, hemorrhagic transformation after cerebral infarction, ICH after thrombolysis, or posterior reversible leukoencephalopathy were excluded. Out of 713 eligible patients with intracerebral hemorrhage from 2005-2012, 287 patients were excluded due to secondary causes of ICH. Of the remaining 426 ICH patients, we excluded those who arrived at our hospital more than 12 hours from ictus, as well as those who did not undergo CTA at admission or at follow-up to determine the factors associated with a spot sign in ICH at the acute stage ( Fig. 1).
Baseline characteristics, laboratory and radiologic profiles
We scrutinized baseline characteristics of the patients including onset age, hypertension, stroke history, antiplatelet drug medication, warfarin medication, reasons for antithrombotics medication, and aggravation of neurologic status indicated by a decrease in the Glasgow Coma Scale (GCS) score Ōēź2 or a decrease in the National Institutes of Health Stroke Scale Ōēź4 within 24 hours of ictus. The hematocrit, international normalized ratio (INR), platelet count, and alanine transaminase (GPT) levels were recorded. Radiologic profiles were also assessed to determine the location of the hemorrhage, volume of the ICH (using the ABC/2 method), location of the spot sign on an enhanced computed tomography (CT) image, intraventricular hemorrhage (IVH) degree (indicating the number of hemorrhage-involved ventricles) and the time from ictus until the CT scan was performed. The location of the hemorrhage was classified as lobar or deep-seated. Hemorrhages originating from the basal ganglia, thalamus, or corpus callosum were categorized as deep-seated hemorrhages. The spot sign was defined as a contrast enhancement focus that was greater than 1 mm in size within the ICH on the axial image of the CTA regardless of the shape of the enhancement ( Fig. 2). Hematoma growth was defined as an absolute increase >6 mL or a relative increase >33% compared with the initial CT image, or an increased IVH determined on a follow-up CT scan within 14 days of ictus.
CT protocol, ICH management, and outcome measurements
All patients presenting with intracranial hemorrhage arrived at our emergency department and underwent brain CT angiography with a multi-detector 64 slice CT scanner (Aquilion, Toshiba Medical System Corporation, Tokyo, Japan). Secondary causes of ICH were thoroughly ruled out through further studies, such as digital subtraction angiography, brain MR imaging, or cerebral venography. A non-contrast CT examination was performed using an axial technique (120 kV, 70 mA, and 5 mm slice thickness reconstruction); a multidetector CT scan was subsequently conducted by scanning from the base of the C7 vertebral body to the vertex using an axial technique (0.641 pitch, 0.5-mm collimation, 350 maximal mA, 120 kVp, 2 mm slice thickness) with 80-100 mL of iodinated contrast material administered using a power injector at 3-4 mL per second into the antecubital vein with an approximately 16- to 18-second delay between the onset of contrast injection and the start of scanning. Protamine sulfate and vitamin K were administered as antidotes to patients with heparin-related and warfarin-related hemorrhage to induce hemostasis. Aminomethylbenzoic acid (150 mg) was routinely injected with normal saline fluid for 3 days. Blood pressure was reduced to <140 mm Hg for systolic blood pressure (SBP) and <90 mm Hg for diastolic blood pressure (DBP) through nicardipine or labetalol IV injections for patients with an initial SBP <180 mm Hg or DBP <110 mm Hg. In the case of an initial SBP >180 mm Hg or DBP >110 mm Hg, the initial SBP or DBP was reduced by approximately 15%. Brain CT or MR scanning was followed-up routinely as follows : 1) between 24 and 72 hours from admission, 2) 4 hours later if a spot sign was found on the initial enhanced CT image, 3) immediately if neurological deterioration occurred, or 4) 7 to 14 days from admission. The hematoma volume was calculated using the formula ABC/2, where A is the greatest diameter on the largest hematoma image, B is perpendicular to line A and C is the vertical depth of the hematoma. The time to reach the target blood pressure according to our center's guideline was measured. The surgical treatment of supratentorial ICH was conducted according to our center's guidelines.
Screening was conducted by a neurological surgeon, and baseline characteristics were investigated by two training residents. Clinical outcomes were assessed by two trained stroke nurses who were not involved in the current study and unaware of the baseline treatments.
Statistics
For the descriptive analysis, all baseline values are displayed as percentage, and the mean values┬▒standard deviation or the median with the interquartile range are shown. Normality tests were performed on all potential continuous variables, and t-tests were conducted to confirm intergroup differences in cases with normal distributions. For categorical variables, a chi-square test was performed between independent two groups. A binary logistic regression method was used to calculate association predictors for the CTA spot sign and hematoma growth. If the p value of a potential factor was less than 0.05 in the univariable analysis, it was entered into the multivariable analysis model, but backward-eliminated to p<0.1. The Hosmer-Lemeshow test was used as a goodness-of-fit test. The Mann-Whitney U test was used to compare variables between two groups with non-normal distributions. p values of less than 0.05 were considered statistically significant. Two-tailed methods were used in all statistical analyses. All statistical calculations were performed using SAS version 9.13 (SAS Institute Inc., Cary, NC).
RESULTS
The mean age of the 287 patients was 61.7 (standard deviation 12.7) years. Of these patients, 161 (56.1%) were male and 126 (43.9%) were female. Hypertension was identified in 151 (52.6%), diabetes in 32 (11.5%), hyperlipidemia or statin medication in 62 (21.6%), current smoking in 103 (35.9%), daily alcohol consumption at an amount >60 g in 41 (14.3%), antiplatelet agent medication in 51 (17.8%), warfarin medication in 7 (2.4%), ischemic stroke history in 11 (3.8%), and hemorrhagic stroke in 6 (2.1%) patients. There were 73 (25.4%) patients with GCS scores of less than 12. Twenty (7.0%) patients deteriorated neurologically within 24 hours of ictus. Forty (13.9%) patients had lobar hemorrhage and 247 (86.1%) had deep-seated hemorrhage. The median hematoma volume was 16 mL (range : 1-160 mL). IVH with or without ICH was noted in 128 (44.6%) patients. The CTA spot sign was identified in 40 (13.9%) patients. Among the 40 patients with the spot sign, 27 (67.5%) had 1 spot, 10 (25%) had 2 spots, 1 (2.5%) had 3 spots, and 2 (5%) had 4 spots. The median Hounsfield units of the spot sign was 152 (range : 74-297). Thirty six (90%) of 40 spot signs were identified within 3 hours from ictus. Hematoma growth within 14 days was identified in 78 (27.2%) patients. Hematoma evacuation was performed in 108 (37.6%) patients. The thirty-day mortality rate was 11.8%. Baseline demography is described in detail in Table 1, 2.
Predictors for CTA spot sign and hematoma growth
In the univariable analysis, reasons for antithrombotics medication, time from ictus to CT scan <3 hours, hemorrhagic volume Ōēź30 mL, and intraventricular extension were significantly associated with a CTA spot sign ( Table 1). These variables, except for antithrombotics medication due to heterogeneous disease entity, were entered into the final multivariable binary logistic regression analysis model ( Table 3). An INR Ōēź1.8, alanine aminotransferase Ōēź40 IU, hemorrhage volume Ōēź30 mL, and a spot sign were significantly associated with hematoma growth ( Table 2) in univariable analysis and then entered into multivariable logistic regression analysis ( Table 4). Warfarin medication and hematocrit were also entered into a multivariable model due to a borderline probability for hematoma growth ( p<0.1) ( Table 2, 4). After adjustment of potential factors, time from ictus to CT scan <3 hours was the only factor identified as a significant predictor for the CTA spot sign (OR, 5.14; 95% CI, 1.76-15.02; p=0.068). Intraventricular extension was revealed as a borderline association factor (OR, 1.96; 95% CI, 0.95-4.05; p=0.002) for the CTA spot sign ( Table 3). On the other hand, a CTA spot sign (OR, 5.70; 95% CI, 2.70-12.01; p<0.001) and elevated GPT level >40 IU (OR, 2.01; 95% CI, 1.01-4.01; p=0.047) were independent predicting factors of hematoma growth. Though an INR of 1.8 or greater was marginally related to hematoma growth in the multivariable binary logistic regression model 1, an INR Ōēź1.8 or warfarin medication was significantly associated with hematoma growth in model 2 ( Table 4). In the subgroup analysis of 78 hematoma growth patients, the time from ictus to hematoma growth was shorter in the group with spot signs than in the group without any spot sign ( Fig. 3). In addition to a spot sign (OR, 3.47; 95% CI, 1.15-10.48; p=0.027) and large hemorrhage volume of 30 mL or more (OR, 3.63; 95% CI, 1.25-10.57; p=0.018), antiplatelet medication (OR, 4.92; 95% CI, 1.31-18.50; p=0.019) showed a significant association with hematoma growth within 6 hours from ictus in multivariable regression analysis ( Table 5).
DISCUSSION
Our study showed that an elapsed time from ictus to CT scan of 3 hours or less was an independent predictor for a CTA spot sign. Additionally, a CTA spot sign and elevated GPT level were independent predictors for hematoma growth of acute ICH patients in Korea. Elevated INR was marginally associated with hematoma growth, and antiplatelet medication was particularly related to hematoma growth within 6 hours of ictus.
Only an elapsed time to CT scan of 3 hours or less was strongly associated with a CTA spot sign in the current study ( Table 3). Similar to our results, an elapsed time to CTA scan of 3 hours or less as well as an intraventricular extension were shown to be predictors of a CTA spot sign by Delgado Almandoz et al. 9,10). However, Radmanesh et al. 33) reported that a hemostatic factor, such as warfarin medication, was a shared predictor for a CTA spot sign for both deep and lobar ICH, while the time to CTA scan was identified as a risk factor only for deep ICH. On the contrary, in the present study, there was no difference in the occurrence of a CTA spot sign between deep and lobar hemorrhages ( Table 1), and intraventricular extension showed a borderline association with the CTA spot sign ( Table 3). In addition, Fig. 3 demonstrates that the time from ictus to hematoma growth of ICH with a CTA spot sign was shorter than that without a CTA spot sign. Furthermore, in hyper-acute ICH within 3 hours of ictus, there may be a high probability of the presence of a CTA spot sign, which leads to early hematoma growth. Many recent studies have reported that a CTA spot sign may be a good radiologic indicator for hematoma growth as well as poor outcome for acute ICH patients in Western countries 9,11,39). Hemostatic factors, such as INR elevation, have been shown in many studies to be risk factors for hematoma growth in acute ICH but not factors in initial ICH volume 5,17,20). The CTA spot sign has also been an independent and strong predictor for hematoma growth and poor outcome 2,9,11,19,25,39). In our results, the presence of a CTA spot sign was also found to be markedly related to hematoma growth and poor outcome ( Table 2, 4). GPT elevation was newly identified as an association factor for hematoma growth. In a large epidemiological study in Asian countries, GPT elevation was revealed to be associated with ICH development in East Asia, and a Korean medical insurance corporation study also showed the association between GPT elevation and ICH occurrence 21,22). Therefore, further research examining GPT elevation and hematoma growth in acute ICH may be necessary. Several previous studies have demonstrated an association between warfarin use and hematoma growth 5,17,20), but the present study did not show a significant association due to testing different populations and a different sampling design. In contrast to the larger percentage of patients who had used warfarin in the prior study (23.0%) 17), our sample population contained a small proportion of only 7 (2.4%) patients who had used warfarin before ICH development; therefore, the effect of warfarin on the size of hematoma growth might be underestimated in our results ( Table 2). One Japanese cohort study was consistent with our finding that anticoagulant medication was not significantly different between patients with and without hematoma growth 36). Moreover, for hemostatic status, an INR elevation of 1.8 or more was a marginally significant predictor of hematoma growth ( Table 4, model 1), and an INR Ōēź1.8 combined with warfarin medication showed a significant association with hematoma growth ( Table 4, model 2). Therefore, a vitamin K antagonist may be a risk factor for hematoma growth in acute ICH. With regard to antiplatelet medication, some investigators have reported increased early hematoma growth, but others could not show that the previous antiplatelet medication was associated with hematoma growth in acute ICH 29,30,35,37). In our results, antiplatelet medication was a significant risk factor for hyper-acute hematoma growth within 6 hours of ictus ( Table 5). An ongoing randomized clinical trial to confirm the efficacy and safety of platelet transfusion in ICH may address this issue 7). To date, the CTA spot sign and contrast extravasation have not been definitively differentiated as early radiologic markers for hematoma growth in acute ICH 2,9,18,39). Moreover, the CTA spot sign has been variously defined as a foci of enhancement within the hematoma on CTA source images (proposed by Wada et al.) 39), and the criteria have included such characteristics as one or more foci of contrast pooling within the ICH, an attenuation of 120 Hounsfield units or more, a discontinuity from normal or abnormal vasculature adjacent to the ICH, as well as having any size or morphology (suggested by Delgado Almandoz et al.) 9). Compared with the rates of CTA spot sign in studies performed in Western countries (22-41%) 11,13,19,39), the spot sign rate in our study (13.9%) was relatively low and in accordance with that of another study in Korea (15%) 32). First, this finding may be attributable to the relatively longer time window of enrollment for patients in our study (as was used in the study by Park et al. 32) and to the inhomogeneous definition of CTA spot sign compared with the results in the literature 9,10,11,19,25,39,40). Second, the first pass CTA may have a lower spot sign detection rate than delayed enhanced CT imaging 23). Prior studies in Western countries have reported that hematoma growth is a strong predictor of early mortality and poor functional outcome 5,6,8,29). In the current study, hematoma growth increased the probability of early death in acute ICH patients ( p=0.018) ( Table 2); this finding is consistent with that of another study in Korea 32). Therefore, hematoma growth may be accepted as one of the important predictors of mortality and poor outcome in Korean patients with acute ICH. The 1-month mortality in this study ( Table 1) was also comparable to those of other studies for Korean patients with acute ICH 24,32). This study revealed the association and risk factors between the CTA spot sign and hematoma growth, as well as the clinical impact for early outcome in Korean patients with acute ICH. However, there were some limitations, including the retrospective study design, multiple testing problems, and a generalizability issue due to the single-center sample.
CONCLUSION
Our results were consistent with those of previous other populations in the way that the CTA spot sign showed a strong probability of being identified in the hyper-acute stage and was an independent strong predictor for hematoma growth in Korean patients with spontaneous ICH. Oral antithrombotics including vitamin K antagonists or antiplatelet drugs might also be associated with hyper-acute hematoma growth in our population. Unlike prior studies, elevated GPT was newly identified as a predictor for hematoma growth and a further prospective study to confirm this association between elevated GPT and hematoma growth in Korean patients with acute ICH.
References
1. Anderson CS, Huang Y, Arima H, Heeley E, Skulina C, Parsons MW, et al : Effects of early intensive blood pressure-lowering treatment on the growth of hematoma and perihematomal edema in acute intracerebral hemorrhage : the Intensive Blood Pressure Reduction in Acute Cerebral Haemorrhage Trial (INTERACT). Stroke 2010, 41 : 307-312,   2. Becker KJ, Baxter AB, Bybee HM, Tirschwell DL, Abouelsaad T, Cohen WA : Extravasation of radiographic contrast is an independent predictor of death in primary intracerebral hemorrhage. Stroke 1999, 30 : 2025-2032,   3. Brouwers HB, Biffi A, McNamara KA, Ayres AM, Valant V, Schwab K, et al : Apolipoprotein E genotype is associated with CT angiography spot sign in lobar intracerebral hemorrhage. Stroke 2012, 43 : 2120-2125,    4. Brouwers HB, Goldstein JN, Romero JM, Rosand J : Clinical applications of the computed tomography angiography spot sign in acute intracerebral hemorrhage : a review. Stroke 2012, 43 : 3427-3432,    5. Cucchiara B, Messe S, Sansing L, Kasner S, Lyden P : CHANT InvestigatorsHematoma growth in oral anticoagulant related intracerebral hemorrhage. Stroke 2008, 39 : 2993-2996,   6. Davis SM, Broderick J, Hennerici M, Brun NC, Diringer MN, Mayer SA, et al : Hematoma growth is a determinant of mortality and poor outcome after intracerebral hemorrhage. Neurology 2006, 66 : 1175-1181,   7. de Gans K, de Haan RJ, Majoie CB, Koopman MM, Brand A, Dijkgraaf MG, et al : PATCH : platelet transfusion in cerebral haemorrhage : study protocol for a multicentre, randomised, controlled trial. BMC Neurol 2010, 10 : 19,    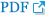 8. Delcourt C, Huang Y, Arima H, Chalmers J, Davis SM, Heeley EL, et al : Hematoma growth and outcomes in intracerebral hemorrhage : the INTERACT1 study. Neurology 2012, 79 : 314-319,   9. Delgado Almandoz JE, Yoo AJ, Stone MJ, Schaefer PW, Goldstein JN, Rosand J, et al : Systematic characterization of the computed tomography angiography spot sign in primary intracerebral hemorrhage identifies patients at highest risk for hematoma expansion : the spot sign score. Stroke 2009, 40 : 2994-3000,    10. Delgado Almandoz JE, Yoo AJ, Stone MJ, Schaefer PW, Oleinik A, Brouwers HB, et al : The spot sign score in primary intracerebral hemorrhage identifies patients at highest risk of in-hospital mortality and poor outcome among survivors. Stroke 2010, 41 : 54-60,   11. Demchuk AM, Dowlatshahi D, Rodriguez-Luna D, Molina CA, Blas YS, Dzialowski I, et al : Prediction of haematoma growth and outcome in patients with intracerebral haemorrhage using the CT-angiography spot sign (PREDICT) : a prospective observational study. Lancet Neurol 2012, 11 : 307-314,   12. Dowlatshahi D, Wasserman JK, Momoli F, Petrcich W, Stotts G, Hogan M, et al : Evolution of computed tomography angiography spot sign is consistent with a site of active hemorrhage in acute intracerebral hemorrhage. Stroke 2014, 45 : 277-280,   13. Ederies A, Demchuk A, Chia T, Gladstone DJ, Dowlatshahi D, Bendavit G, et al : Postcontrast CT extravasation is associated with hematoma expansion in CTA spot negative patients. Stroke 2009, 40 : 1672-1676,   14. Emlet LL, Crippen D : Early recombinant activated factor VII for intracerebral hemorrhage reduced hematoma growth and mortality, while improving functional outcomes. Crit Care 2006, 10 : 304,    15. Feigin VL, Lawes CM, Bennett DA, Barker-Collo SL, Parag V : Worldwide stroke incidence and early case fatality reported in 56 population-based studies : a systematic review. Lancet Neurol 2009, 8 : 355-369,   16. Flaherty ML, Woo D, Haverbusch M, Sekar P, Khoury J, Sauerbeck L, et al : Racial variations in location and risk of intracerebral hemorrhage. Stroke 2005, 36 : 934-937,   17. Flibotte JJ, Hagan N, O'Donnell J, Greenberg SM, Rosand J : Warfarin, hematoma expansion, and outcome of intracerebral hemorrhage. Neurology 2004, 63 : 1059-1064,   18. Goldstein JN, Fazen LE, Snider R, Schwab K, Greenberg SM, Smith EE, et al : Contrast extravasation on CT angiography predicts hematoma expansion in intracerebral hemorrhage. Neurology 2007, 68 : 889-894,   19. Hallevi H, Abraham AT, Barreto AD, Grotta JC, Savitz SI : The spot sign in intracerebral hemorrhage : the importance of looking for contrast extravasation. Cerebrovasc Dis 2010, 29 : 217-220,   20. Huttner HB, Schellinger PD, Hartmann M, K├Čhrmann M, Juettler E, Wikner J, et al : Hematoma growth and outcome in treated neurocritical care patients with intracerebral hemorrhage related to oral anticoagulant therapy : comparison of acute treatment strategies using vitamin K, fresh frozen plasma, and prothrombin complex concentrates. Stroke 2006, 37 : 1465-1470,   21. Kim HC, Kang DR, Nam CM, Hur NW, Shim JS, Jee SH, et al : Elevated serum aminotransferase level as a predictor of intracerebral hemorrhage : Korea medical insurance corporation study. Stroke 2005, 36 : 1642-1647,   22. Kim HC, Oh SM, Pan WH, Ueshima H, Gu D, Chuang SY, et al : Association between alanine aminotransferase and intracerebral hemorrhage in East Asian populations. Neuroepidemiology 2013, 41 : 131-138,   23. Kim J, Smith A, Hemphill JC 3rd, Smith WS, Lu Y, Dillon WP, et al : Contrast extravasation on CT predicts mortality in primary intracerebral hemorrhage. AJNR Am J Neuroradiol 2008, 29 : 520-525,    24. Kim KH : Predictors of 30-day mortality and 90-day functional recovery after primary intracerebral hemorrhage : hospital based multivariate analysis in 585 patients. J Korean Neurosurg Soc 2009, 45 : 341-349,    25. Li N, Wang Y, Wang W, Ma L, Xue J, Weissenborn K, et al : Contrast extravasation on computed tomography angiography predicts clinical outcome in primary intracerebral hemorrhage : a prospective study of 139 cases. Stroke 2011, 42 : 3441-3446,   26. Mart├Ł-F├Ābregas J, Borrell M, Silva Y, Delgado-Mederos R, Mart├Łnez-Ram├Łrez S, de Juan-Delago M, et al : Hemostatic proteins and their association with hematoma growth in patients with acute intracerebral hemorrhage. Stroke 2010, 41 : 2976-2978,   27. Mayer SA, Brun NC, Begtrup K, Broderick J, Davis S, Diringer MN, et al : Efficacy and safety of recombinant activated factor VII for acute intracerebral hemorrhage. N Engl J Med 2008, 358 : 2127-2137,   28. Mendelow AD, Gregson BA, Rowan EN, Murray GD, Gholkar A, Mitchell PM, et al : Early surgery versus initial conservative treatment in patients with spontaneous supratentorial lobar intracerebral haematomas (STICH II) : a randomised trial. Lancet 2013, 382 : 397-408,    29. Moussouttas M, Malhotra R, Fernandez L, Maltenfort M, Holowecki M, Delgado J, et al : Role of antiplatelet agents in hematoma expansion during the acute period of intracerebral hemorrhage. Neurocrit Care 2010, 12 : 24-29,   30. Naidech AM, Jovanovic B, Liebling S, Garg RK, Bassin SL, Bendok BR, et al : Reduced platelet activity is associated with early clot growth and worse 3-month outcome after intracerebral hemorrhage. Stroke 2009, 40 : 2398-2401,   31. Park HS, Kang MJ, Huh JT : Recent epidemiological trends of stroke. J Korean Neurosurg Soc 2008, 43 : 16-20,    32. Park SY, Kong MH, Kim JH, Kang DS, Song KY, Huh SK : Role of 'spot sign' on CT angiography to predict hematoma expansion in spontaneous intracerebral hemorrhage. J Korean Neurosurg Soc 2010, 48 : 399-405,    33. Radmanesh F, Falcone GJ, Anderson CD, Battey TW, Ayres AM, Vashkevich A, et al : Risk factors for computed tomography angiography spot sign in deep and lobar intracerebral hemorrhage are shared. Stroke 2014, 45 : 1833-1835,    34. Rodriguez-Luna D, Pi├▒eiro S, Rubiera M, Ribo M, Coscojuela P, Pagola J, et al : Impact of blood pressure changes and course on hematoma growth in acute intracerebral hemorrhage. Eur J Neurol 2013, 20 : 1277-1283,   35. Sansing LH, Messe SR, Cucchiara BL, Cohen SN, Lyden PD, Kasner SE, et al : Prior antiplatelet use does not affect hemorrhage growth or outcome after ICH. Neurology 2009, 72 : 1397-1402,    36. Takeda R, Ogura T, Ooigawa H, Fushihara G, Yoshikawa S, Okada D, et al : A practical prediction model for early hematoma expansion in spontaneous deep ganglionic intracerebral hemorrhage. Clin Neurol Neurosurg 2013, 115 : 1028-1031,   37. Toyoda K, Okada Y, Minematsu K, Kamouchi M, Fujimoto S, Ibayashi S, et al : Antiplatelet therapy contributes to acute deterioration of intracerebral hemorrhage. Neurology 2005, 65 : 1000-1004,   38. van Asch CJ, Luitse MJ, Rinkel GJ, van der Tweel I, Algra A, Klijn CJ : Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin : a systematic review and meta-analysis. Lancet Neurol 2010, 9 : 167-176,   39. Wada R, Aviv RI, Fox AJ, Sahlas DJ, Gladstone DJ, Tomlinson G, et al : CT angiography "spot sign" predicts hematoma expansion in acute intracerebral hemorrhage. Stroke 2007, 38 : 1257-1262,   40. Wang YH, Fan JY, Luo GD, Lin T, Xie DX, Ji FY, et al : Hematoma volume affects the accuracy of computed tomographic angiography 'spot sign' in predicting hematoma expansion after acute intracerebral hemorrhage. Eur Neurol 2011, 65 : 150-155,  
Fig.┬Ā1
Flowchart of patient selection. ICH : intracerebral hemorrhage, AVM : arteriovenous malformation, AVF : arteriovenous fistula. 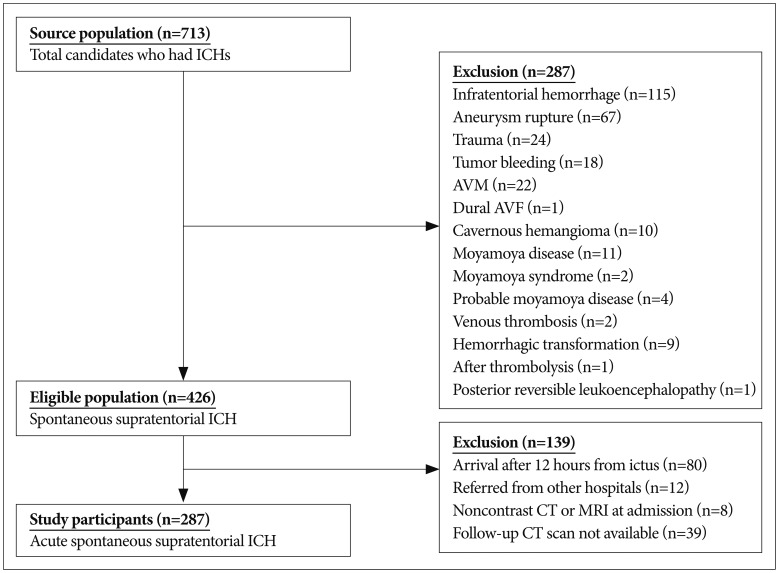
Fig.┬Ā2
Representative CT angiography (CTA) images obtained from a 50-year-old woman with a history of daily alcohol consumption who arrived at our hospital within 42 minutes of ictus. A : Non-contrast brain CT showing an acute hemorrhage in the right putamen. B : A small enhanced focus of contrast in the middle of the hematoma (black arrow). C : CTA image showing no intracranial vessel abnormality. D : Hematoma growth identified on a brain CT image acquired immediately after her neurologic deterioration 4 hours after arrival at our hospital. 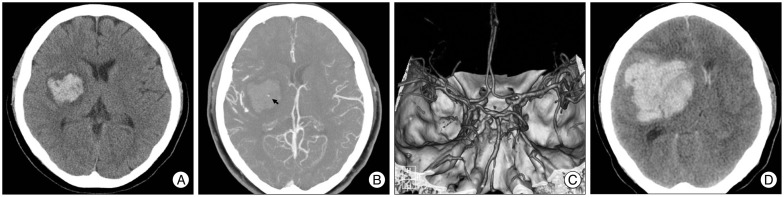
Fig.┬Ā3
Among the 78 patients with hematoma growth, a comparison of the time from ictus to hematoma growth between the spot sign-positive group (n=25, 32.1%) and the spot sign-negative group (n=53, 67.9%). A Mann-Whitney U test was used to compare the two groups. 
Table┬Ā1
Clinical, laboratory, and radiologic characteristics of acute ICH patients with or without CTA spot sign 
Table┬Ā2
Clinical, laboratory, and radiologic characteristics of acute ICH patients with or without hematoma growth 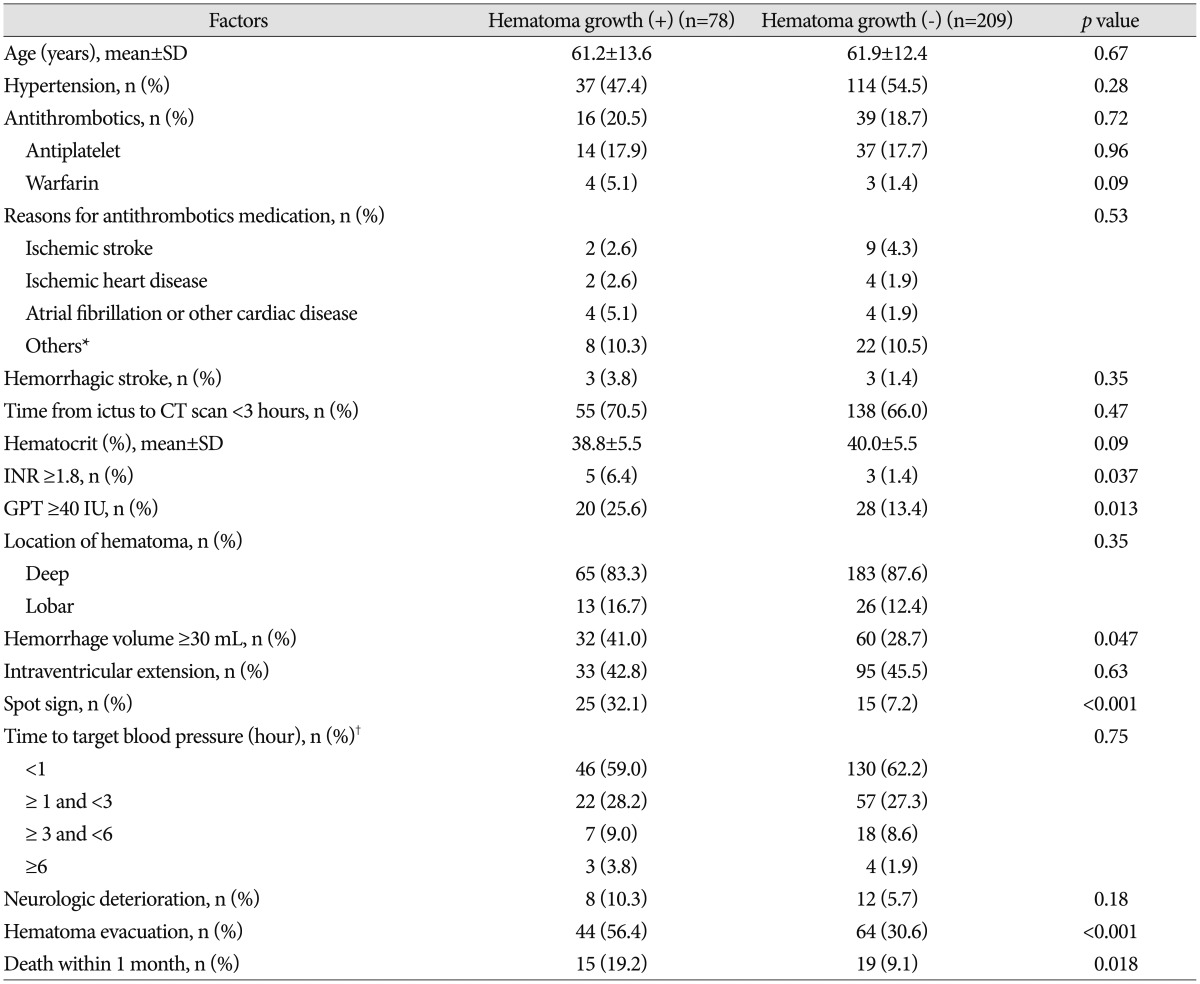
Table┬Ā3
Predictors of CTA spot sign in acute spontaneous ICH 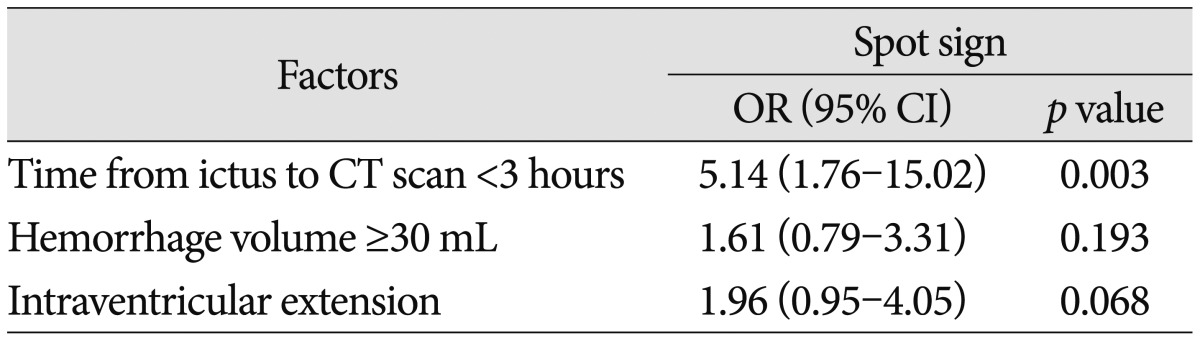
Table┬Ā4
Predictors of hematoma growth in acute spontaneous ICH 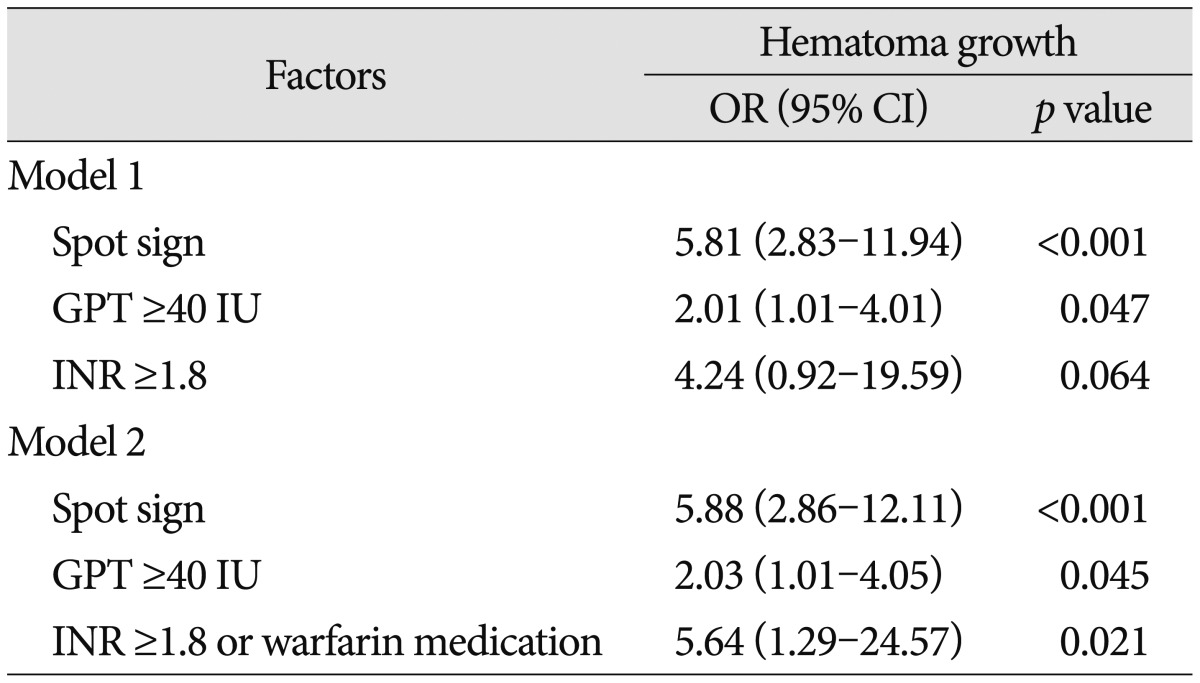
Table┬Ā5
Predictors of hematoma expansion within 6 hours in 78 hematoma growth patients 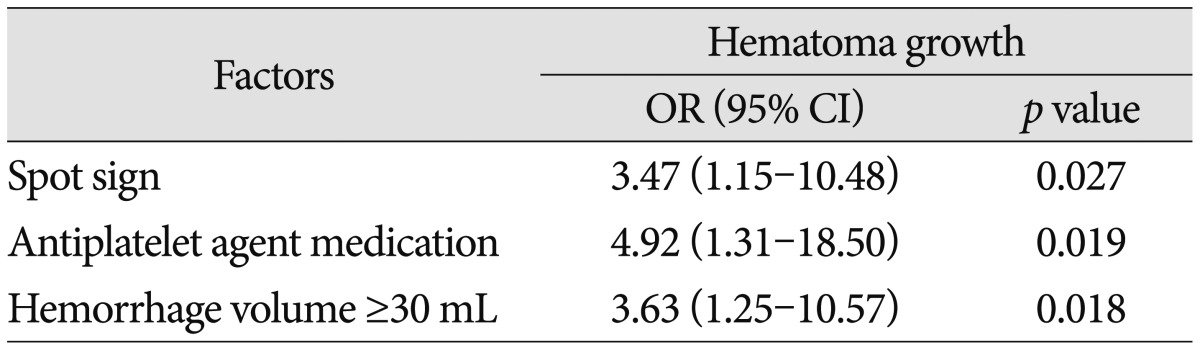
|
|























