Sun, Tao, Luo, Gao, and Shang: Type 1.5 Split Cord Malformation : A New Theory of Pathogenesis
Abstract
To report two cases of type 1.5 split cord malformation (SCM), a subtype of SCM with combined characteristics of types I and II and to review the relevant literature and propose a new possible pathogenetic theory for type 1.5 SCM. A 52-year-old woman had hemicords within a single dural sac with a dorsal bony septum at the L5 level. A 9-year-old boy had hemicords within a single dural sac with a ventral bony septum and fibrous extension at the L3 level. Both patients underwent microsurgical treatments for removing the bony septum, detethering the spinal cord, and sectioning the filum terminale. The surgical procedure revealed an extradural partial bony septum and hemicords within an intact single dural sac in each patient. Both patients were discharged from the hospital without de novo nerve dysfunction. Published cases have validated that types I and II SCM can overlap. We recommend recent type 1.5 SCM as a normative terminology for this overlapping SCM and report two rare cases of this SCM. We propose an associated pathogenesis consisting of uneven distribution and regression to explain type 1.5 SCM. Furthermore, we postulate that the amount of condensing meninx primitiva might determine whether the left bony septum has fibrous extensions to the opposite dura in type 1.5 SCM.
Key Words: Split cord malformation · Pathogenesis · Embryogenesis · Meninx · Regression.
INTRODUCTION
Split cord malformation (SCM), a congenital neural tube defect, presents with longitudinally separated functional hemicords. Pang et al. [ 16] classified SCM into two types. In type I SCM (diastematomyelia), the hemicords are divided by a bony sagittal septum within double dural sacs. In type II SCM (diplomyelia), the hemicords are divided by a thin sagittal fibrous septum within a single dural sac [ 16]. However, some authors have reported a few cases demonstrating characteristics of both type I and type II SCM, that is, mixed, intermediate, or composite type SCM [ 2, 11, 18]. Recently, Meena et al. [ 15] suggested type 1.5 SCM to define these unusual cases. In this report, we present two cases of type 1.5 SCM and review the relevant literature. We also discuss the previously hypothesized pathogenesis and propose a new possible pathogenetic theory.
CASE REPORT
Case 1
A 52-year-old woman had been suffering from constipation since 2012. She experienced pain in the dorsum of both lower limbs (more severe on the left) and numbness of the left foot in 2019. Standing up after sedentariness induced these symptoms. Uroschesis occurred at the end of 2019. The patient was admitted to our department in July 2020. A physical examination revealed a lumbosacral dermal sinus with central hyperpigmentation and tenderness ( Fig. 1A), a weakened anal reflex, and intact sensory functions. Muscle strength of the four limbs was grade 5/6. Magnetic resonance imaging (MRI) demonstrated a dorsal bulging lesion at the L5 level, hemicords within a single dural sac, and a low conus at the S1 level ( Fig. 1B- D). Computed tomography (CT) revealed a dorsal bony septum at the L5 level ( Fig. 2A- C). The patient underwent a single-stage microsurgery for resecting the dermal sinus and the bony septum, detethering the spinal cord, and sectioning the filum terminale. During the surgical procedure, we found a sinus tract communicating with the intradural space, a dorsal bony septum pointing to the midline of the dural sac without penetrating it (an intact single dural sac) ( Fig. 2D), and a thickened and tight filum terminale. No fibrous extension from the bony septum to the ventral dura was noted. The patient was discharged from the hospital 14 days postoperatively without de novo nerve dysfunction.
Case 2
A 9-year-old boy was found to have asymmetric shoulders (higher on the right) and daily urine suppression since March 2020. He was diagnosed with scoliosis and butterfly vertebra at the T4 level by radiography and CT examinations at a local hospital. He was admitted to our department in August 2020 for surgical treatment. A physical examination revealed a wrinkled lumbosacral cutaneous pit without hair. Preoperative MRI demonstrated a ventral spinous lesion at the L3 level, hemicords within a single dural sac, and a low conus at the L5 level ( Fig. 3). CT identified a ventral bony septum at the L3 level ( Fig. 4A- C). Three-dimensional CT reconstruction indicated lumbosacral spina bifida ( Fig. 4D). We performed a single-stage microsurgery to resect the bony septum, detether the spinal cord, and section the filum terminale. The surgical procedure revealed hemicords within a single dural sac ( Fig. 5A), a ventral bony septum with fibrous extension to the dorsal dura ( Fig. 5B and C), and a filum terminale lipoma. The bony septum and fibrous extension were completely resected ( Fig. 5D). The patient was discharged from the hospital 12 days postoperatively without de novo nerve dysfunction.
DISCUSSION
We presented two different cases of type 1.5 SCM in this report. Due to the rarity of type 1.5 SCM, these two cases are rather valuable. Furthermore, we reviewed the pertinent literature to cover detailed information of published reports, which could help neurosurgeons obtain more knowledge regarding type 1.5 SCM.
SCM is a congenital abnormality, within the spectrum of neural tube defects, in which a bony or fibrous septum longitudinally divides the spinal cord into two parts. Due to distinct conceptions, previous authors named SCM with confusing terminology. Diastematomyelia was adopted by Hertwig [ 10] and supported by some other authors [ 12, 14], while Bruce et al. [ 4] proposed diastematomyelia for the hemicords with a midline bony spur and diplomyelia for the hemicords without the spur. Other authors, however, preferred diplomyelia regardless of the septal components [ 9, 13, 17]. In 1992, Pang et al. [ 16] proposed a novel nomenclature that classified SCM into two types : type I SCM (formerly diastematomyelia), presenting with hemicords within double dural sacs split by a bony sagittal septum and type II SCM (formerly diplomyelia), in which hemicords lie in an intact single dural sac with a thin midline sagittal fibrous septum. They also postulated a unified theory to elucidate the pathogenesis of SCM during embryogenesis [ 16]. According to the unified theory, both types result from a common ontogenetic error [ 16]. The meninx primitiva appears over the ventral neural tube and migrates dorsally to wrap it, forming the dural sac. If an abnormal midline fistula of the primitive dura mater forms simultaneously with the normal primitive neurenteric canal (at approximately 21 days’ gestational age), the mesenchyme will condense around the fistula, form an endomesenchymal tract, and recruit the meninx primitiva into the tract (around 30 days’ gestational age). It is this meninx primitiva that differentiates into the future dura mater and bony septum, and type I SCM develops. If the endomesenchymal tract appears earlier than day 21, the mesenchyme will not collect the meninx primitiva. This results in a thin fibrous septum distinguishable from normal dura mater between the hemicords or type II SCM. Pang et al. [ 16] believed that the unified theory was validated by the fact that all 39 patients with SCM in their series did not manifest combined features of types I and II. However, some authors have reported a few cases with characteristics of both types I and II SCM, meaning that the lesion incorporated the hemicords within a single dural sac and an extradural partial bony septum [ 1, 3, 5, 7, 8, 15, 18, 19, 21]. We reviewed the literature, and 13 pertinent cases are listed in Table 1. There is a lack of consensus on the terminology for this combined SCM, whose terminology includes mixed SCM [ 18], intermediate SCM [ 11], and composite SCM [ 16]. Recently, Meena et al. [ 15] proposed type 1.5 SCM to define this unusual variation. Based on the location of the bony septum, they classified type 1.5 SCM into two subtypes : type 1.5a SCM, with the vertebral plate arising, and type 1.5b SCM, with the vertebral body arising [ 15]. We believe that this simplified terminology adequately describes the characteristics of this unusual SCM. Referring to this method, we classified our case 1 as type 1.5a SCM and case 2 as type 1.5b SCM. We hope that this terminology will be adopted by more authors and applied in future studies to unify the nomenclature. Different authors have held various views to explain the pathogenesis of type 1.5 SCM. Vaishya and Kumarjain [ 20] supported the unified theory and considered that all variants of SCM shared a common pathogenesis in which the proportion of the meninx primitiva was the only influencing factor determining the SCM type : the more meninx primitiva cells present in the endomesenchymal tract, the more complete is bony septum and dura formation. Chandra et al. [ 5] reported a case of dorsal bony septum originating from the vertebral arch. They questioned the unified theory in which the bony septum should develop from the vertebral body where the migration of the meninx primitiva begins [ 16], and hypothesized that the meninx primitiva first migrates along the lateral side of the hemicords, accumulates dorsally, and returns along the endomesenchymal tract dorsoventrally, then forms a dorsal bony septum [ 5]. In the unified theory, the bony septum and hypertrophic fusion of adjacent laminae should co-exist at the same level in type I SCM, because the meninx primitiva in the endomesenchymal tract could simultaneously induce both lesions [ 16]. Salunke et al. [ 18] reported two cases of type I SCM with normal laminae; hence, they doubted the value of the meninx primitiva in forming the bony septum and pointed out a possible role of the pluripotent mesenchyme. They also surmised that the regression of the endomesenchymal tract might cause type 1.5 SCM [ 18]. To explain the formation of all subtypes of type 1.5 SCM described in previous reports and our cases, we postulate an associative pathogenesis of type 1.5 SCM, consisting of uneven distribution of meninx primitiva and regression of the bony septum. In case 1, we intraoperatively found a dorsal bony septum at the L5 level and fused laminae of L4 and L5. Case 2, however, had normal laminae opposite to the ventral bony septum. We conjecture that uneven distribution of the meninx primitiva between the hemicords might cause less cells of the meninx primitiva to mix with the neural arch cells, which weakens the sclerosing effect [ 2], resulting in normal laminae without hypertrophic fusion. This uneven distribution could also explain Salunke et al.’s cases [ 18]. Case 2 had a fibrous extension from the bony septum to the opposite dura, but case 1 did not. We hypothesize the regression of the bony septum as a possible mechanism for this left fibrous extension because some studies have validated that regression plays a key role in neurulation and neural tube development [ 6, 22]. In other words, type 1.5 SCM might be derived from type I SCM. In type I SCM, with uneven distribution, less meninx primitiva forms a less stable part of the bony septum. With some unknown factors, this unsubstantial part might regress, leaving a half bony septum. In case 2, we consider that relatively fewer cells of the meninx primitiva near the neural arch formed neither hypertrophic laminae nor a solid dorsal bony septum. Some factors might drive the dorsal part to regress into a fibrous extension ( Fig. 6A). In case 1, it is possible that few meninx primitiva cells exist around the vertebral body, resulting in a remnant dorsal bony septum without any ventral fibrous extension ( Fig. 6B), which could also explain the case presented by Chandra et al. [ 5]. Therefore, we postulate that the presence of a fibrous extension might depend on the amount of condensing meninx primitiva. Considering that type 1.5 SCM is extremely rare, we regard regression to be accidental. Pang et al. [ 16] reported that type II SCM was more frequent than type I SCM (around 60% and 40%, respectively). Hence, we believe that type II SCM in which only fibrous septa exist should not regress from type I SCM. The pathogenesis of type I and II SCM defined in the unified theory should be conceivable, and our associative theory is only responsible for type 1.5 SCM. To explicitly describe the true mechanisms, an experimental model of embryogenesis may be necessary in the future.
CONCLUSION
In addition to the unified theory, types I and II SCM could overlap. We recommend type 1.5 SCM, as proposed by Meena et al. [ 15], as a normative terminology for this overlapping SCM. We hypothesize an associated pathogenesis consisting of uneven distribution and regression to explain type 1.5 SCM. Furthermore, we postulate that the amount of condensing meninx primitiva might determine whether the bony septum has a fibrous extension to the opposite dura in type 1.5 SCM. We believe that the application of type 1.5 SCM could provide a better classification for neurosurgeons to clarify the type, and the novel possible pathogenesis of type 1.5 SCM may provide a new idea for revealing the origin of SCM.
Fig. 1.
The lumbosacral dermal sinus (about 4×4 cm) with central hyperpigmentation (A). Magnetic resonance imaging suggesting a dorsal bulging lesion (arrowhead) at the L5 level and a low conus at the S1 level (B : axial T1-weighted sequences; C : sagittal T1-weighted sequences; D : sagittal T2-weighted sequences). 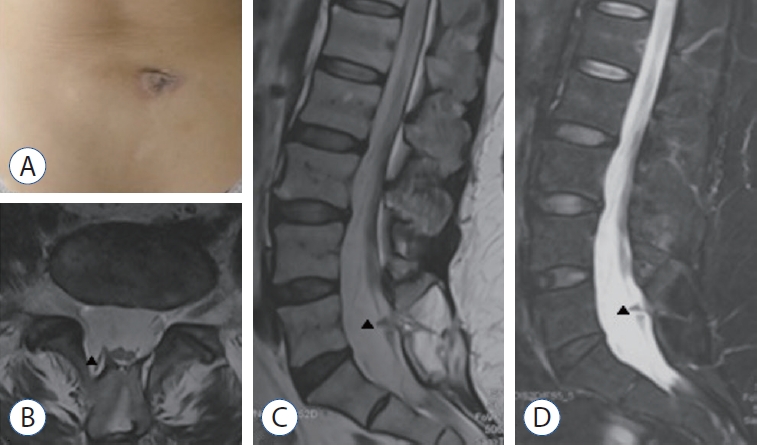
Fig. 2.
Computed tomography suggesting a dorsal bony septum (asterisk) at the L5 level (A : sagittal view; B : coronal view; C : axial view). A partially resected dorsal bony septum (asterisk) found intraoperatively. Its base was sectioned during the primary laminectomy (D). 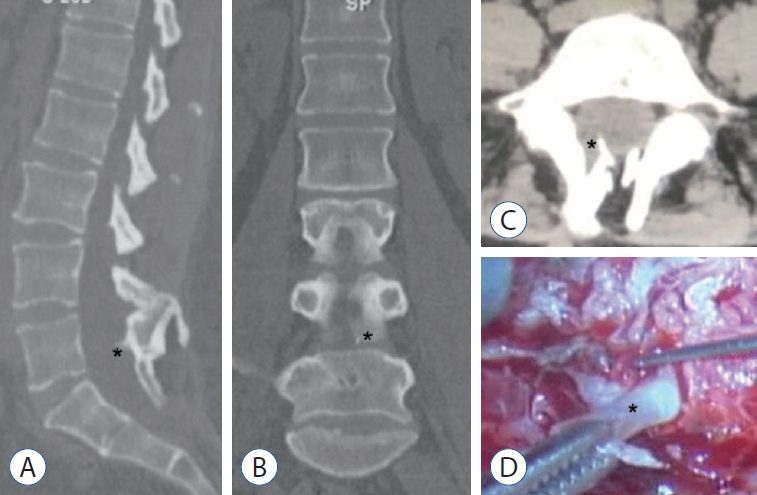
Fig. 3.
Magnetic resonance imaging suggesting a ventral spinous lesion (arrow) at the L3 level and a low conus at the L5 level (A : sagittal T1-weighted sequences; B : sagittal T2-weighted sequences; C: axial T2-weighted sequences). Split spinal cord (D). 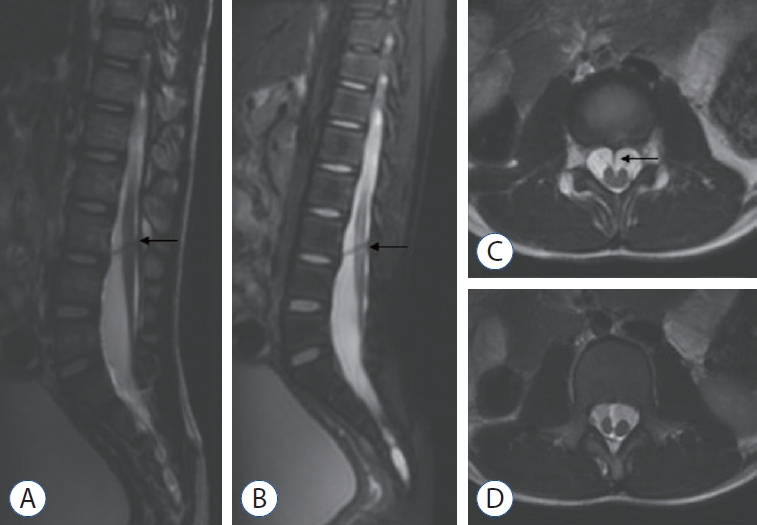
Fig. 4.
Computed tomography (CT) demonstrating a ventral bony septum (dotted arrow) at the L3 level (A : sagittal view; B : axial soft-tissue window; C : axial bone window). Three-dimensional CT reconstruction indicating lumbosacral spina bifida (D). 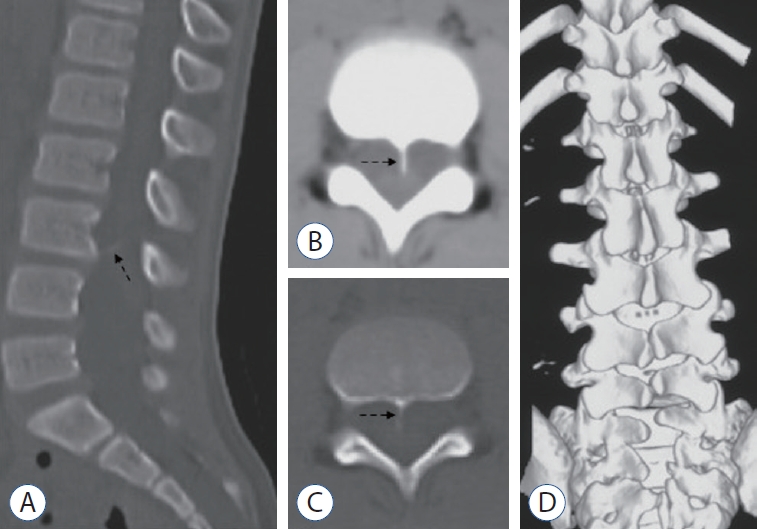
Fig. 5.
A : An intact single dural sac. B : A fibrous extension to the dorsal dura (pound). C : A ventral bony septum (cross). D : Complete resection of the bony septum. 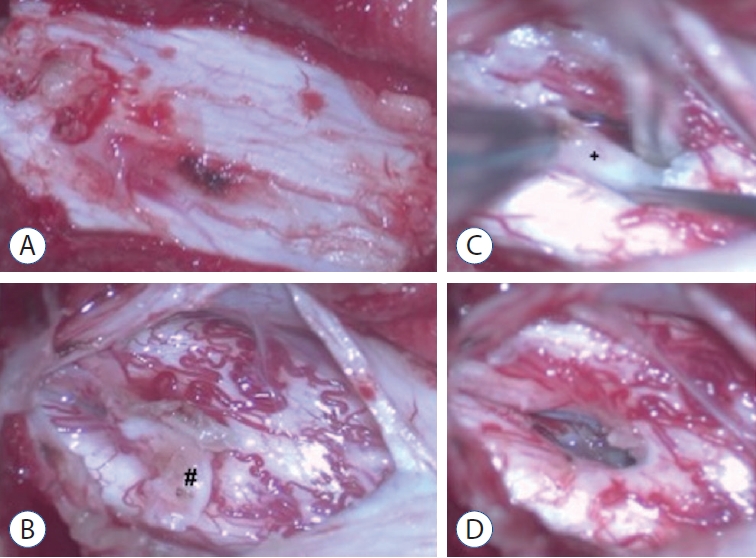
Fig. 6.
A relatively lower proportion of meninx primitiva near the neural arch regressing into a fibrous extension from the ventral bony septum in case 2 (A). Few meninx primitiva cells existing around the vertebral body, resulting in a regressed dorsal bony septum without any ventral fibrous extension in case 1 (B). 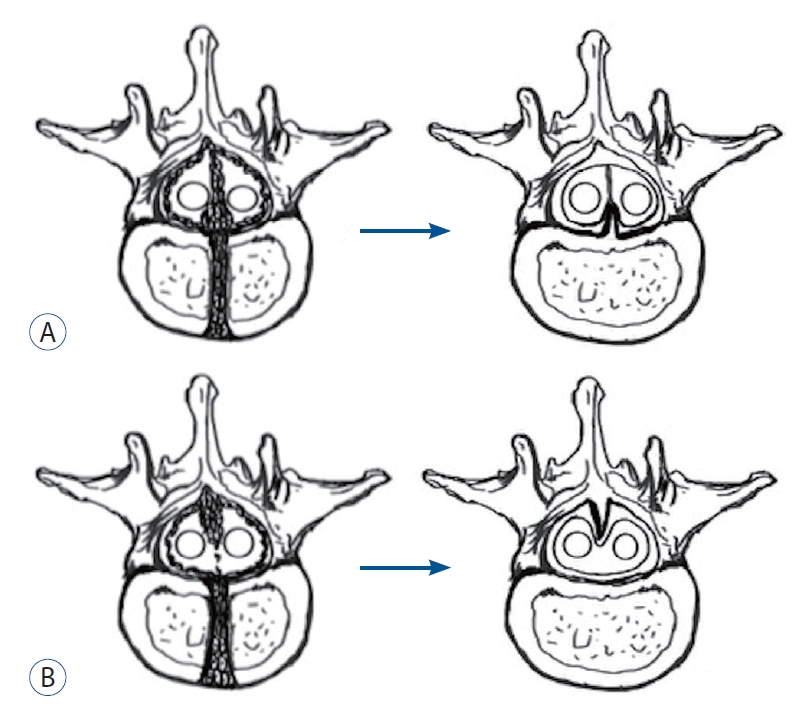
Table 1.
|
Study |
Age (years)/sex |
Clinical presentation |
Radiological presentation |
Intraoperative presentation |
|
Chandra et al. [5] (1999) |
9/F |
Right-sided sprengel shoulder deformity |
A dorsal bony septum at L2-3 |
Thick filum terminale |
|
Lumbosacral hair |
Split cord beginning at L2 |
No fibrous extension |
|
Tethered cord at L4 |
|
Low conus at L5 |
|
Erşahin [7] (2000) |
14 months/M |
Lumbar hypertrichosis |
A dorsal bony septum at L3 |
Tight filum terminale |
|
Widened spinal canal |
Fibrous bands and dorsal paramedian roots between the dorsal surface of hemicords and the dorsal dura |
|
Bifid laminae at L5-S1 |
No fibrous extension |
|
Basak et al. [3] (2002) |
3/F |
Hair-covered mass of the upper dorsal spine |
A dorsal bony septum at C5-6 |
No fibrous extension |
|
Split cord at C3-T1 |
|
Syrinx cavities at C3-T1and T3-T7 |
|
Akay et al. [1] (2002) |
7/F |
Lumbar hair |
A dorsal bony septum at L4 |
Rootlets coming out from the medial aspect of the hemicords |
|
Low conus at L4 |
No fibrous extension |
|
Akay et al. [1] (2002) |
2/F |
A sacral rigid mass lesion |
A dorsal bony septum at S1 |
Rootlets coming out from the dorsal medial aspect of the hemicords |
|
A bifid lamina at S1 |
Thick filum terminale |
|
Conus medullaris at coccyx |
Fibrous ligaments lying on the dorsal dura |
|
No fibrous extension |
|
Van Aalst et al. [21] (2005) |
15/M |
Walking difficulties |
A dorsal bony septum at L3-L4 |
Arachnoidal adhesions between the hemicords |
|
Pain in the lower extremities, scoliosis |
Fibrous extension to the ventral dura |
|
Singh et al. [19] (2011) |
1/F |
Hair over the middorsal spine with hyperpigmentation |
A bony septum at T5-T9 |
Hemicords with separate dura sacs at T2-L1 |
|
A ventral bony septum at T11 |
Hemicords with a single dura sac at T11 |
|
Split cord at T2-L1 |
Thick filum terminale |
|
Low conus at L3-4 |
No fibrous extension |
|
Salunke et al. [18] (2011) |
7/M |
A lipomatous swelling |
A ventral bony septum at lower dorsal level |
The paramedian nerve roots attaching to one another |
|
Lumbar hypertrichosis |
Bifid laminae |
Sinus tract extending extradurally |
|
Weakness of the left lower limb |
No fibrous extension |
|
Salunke et al. [18] (2011) |
1/M |
A small midline swelling at the lower back |
A lumbosacral dorsal bony septum |
Fibrous extension to the ventral dura |
|
Low conus to the sacrum |
|
Garg et al. [8] (2015) |
10 months/F |
Lumbar midline hair |
A ventral bony septum at L2-L3 |
Thick filum terminale |
|
Lower limbs moving less |
Lumbosacral spinal dysraphism |
No fibrous extension |
|
Scoliosis |
Low conus at L3 |
|
Meena et al. [15] (2020) |
11/M |
Low back pain |
A ventral bony septum at T12-L1 |
Thick filum terminale |
|
Split cord from T11 to the cord ending |
No fibrous extension |
|
Low conus at L3-L4 |
|
Hypoplastic T10-L2 vertebral body |
|
Present case 1 |
52/F |
A lumbosacral dermal sinus with central hyperpigmentation and tenderness |
A dorsal bony septum at L5 |
Sinus tract communicated with intradural space |
|
Pain on the dorsal side of both lower limbs |
Low conus at S1 |
Thick and tight filum terminale |
|
Numbness of the left foot |
Lumbosacral spina bifida |
No fibrous extension |
|
Present case 2 |
9/M |
Asymmetric shoulders |
A ventral bony septum |
Filum terminale lipoma |
|
Scoliosis |
Low conus at L5 |
Fibrous extension to the dorsal dura |
|
A lumbosacral wrinkled cutaneous pit |
Butterfly vertebra at T4 |
|
Urine suppression |
Lumbosacral spina bifida |
References
1. Akay KM, Izci Y, Baysefer A : Dorsal bony septum: a split cord malformation variant. Pediatr Neurosurg 36 : 225-228, 2002   2. Akay KM, Izci Y, Baysefer A, Timurkaynak E : Composite type of split cord malformation: two different types at three different levels: case report. J Neurosurg 102( 4 Suppl):436-438, 2005   3. Basak M, Ozel A, Erturk M : An unusual case of diastematomyelia. Presence of one dural sheath associated with a bony spur. Acta Radiol 43 : 626, 2002   4. Bruce A, M’Donald S, Pirie JHH : A second case of partial doubling of the spinal cord. Rev Neurol Psychiatr 4 : 6, 1906
5. Chandra PS, Kamal R, Mahapatra AK : An unusual case of dorsally situated bony spur in a lumbar split cord malformation. Pediatr Neurosurg 31 : 49-52, 1999   6. Colas JF, Schoenwolf GC : Towards a cellular and molecular understanding of neurulation. Dev Dyn 221 : 117-145, 2001   7. Erşahin Y : An unusual split cord malformation. Pediatr Neurosurg 32 : 109, 2000   8. Garg K, Mahapatra AK, Tandon V : A rare case of type 1 C split cord malformation with single dural sheath. Asian J Neurosurg 10 : 226-228, 2015    9. Herren RY, Edwards JE : Diplomyelia (duplication of the spinal cord). Arch Pathol 30 : 1203-1213, 1940
10. Hertwig O : Urmund und spina bifida. Archiv f mikrosk Anatomie 39 : 353-502, 1892  11. Izci Y, Kural C : Composite type of split cord malformation: rare and difficult to explain. Pediatr Neurosurg 47 : 461, 2011   12. Lichtenstein BW : Spinal dysraphism: spina bifida and myelodysplasia. Arch Neur Psych 44 : 792-810, 1940
13. Marr GE, Uihlein A : Diplomyelia and compression of the spinal cord and not of the cauda equina, by a congenital anomaly of the third lumbar vertebra. Surg Clin North Am 24 : 963-977, 1944
14. Maxwell HP, Bucy PC : Diastematomyelia; report of a clinical case. J Neuropathol Exp Neurol 5 : 165-167, 1946   15. Meena RK, Doddamani RS, Gurjar HK, Kumar A, Chandra PS : Type 1.5 split cord malformations: an uncommon entity. World Neurosurg 133 : 142-149, 2020   16. Pang D, Dias MS, Ahab-Barmada M : Split cord malformation: part I: a unified theory of embryogenesis for double spinal cord malformations. Neurosurgery 31 : 451-480, 1992  17. Pickles W : Duplication of the spinal cord; an account of a clinical example with a consideration of other reports. J Neurosurg 6 : 324-331, 1949  18. Salunke P, Kovai P, Malik V, Sharma M : Mixed split cord malformation: are we missing something? Clin Neurol Neurosurg 113 : 774-778, 2011   19. Singh PK, Khandelwal A, Singh A, Ailawadhi P, Gupta D, Mahapatra AK : Long-segment type 1 split cord malformation with two-level split cord malformation and a single dural sac at the lower split. Pediatr Neurosurg 47 : 227-229, 2011   20. Vaishya S, Kumarjain P : Split cord malformation: three unusual cases of composite split cord malformation. Childs Nerv Syst 17 : 528-530, 2001   21. Van Aalst J, Beuls EA, Vles JS, Cornips EM, van Straaten HW : The intermediate type split cord malformation: hypothesis and case report. Childs Nerv Syst 21 : 1020-1024, 2005   22. Xu X, Li C, Takahashi K, Slavkin HC, Shum L, Deng CX : Murine fibroblast growth factor receptor 1alpha isoforms mediate node regression and are essential for posterior mesoderm development. Dev Biol 208 : 293-306, 1999  
|
|






















